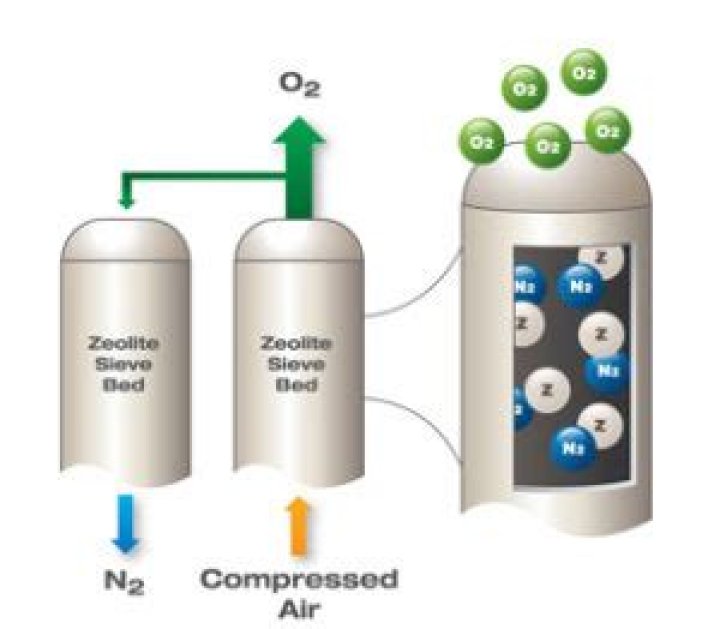
What is an adsorber?
Solids that are used to adsorb gases or dissolved substances are called adsorbents; the adsorbed molecules are usually referred to collectively as the adsorbate. An example of an excellent adsorbent is the charcoal used in gas masks to remove poisons or impurities from a stream of air.
What do adsorption isotherms show?
An adsorption isotherm is a graph that represents the variation in the amount of adsorbate(x) adsorbed on the surface of the adsorbent with the change in pressure at a constant temperature.
What is adsorption capacity?
Adsorption capacity (or loading) is the amount of adsorbate taken up by the adsorbent per unit mass (or volume) of the adsorbent.
What is adsorption article?
Adsorption is a mass transfer process that is a phenomenon of sorption of gases or solutes by solid or liquid surfaces. The adsorption on the solid surface is that the molecules or atoms on the solid surface have residual surface energy due to unbalanced forces.
What is adsorber vessel?
Adsorber vessels are designed to dissolve high concentrations of gaseous components into liquids. Activated carbon adsorption is an effective technology for removal of contaminants from both air and liquids. HVM can fabricate and manufacture activated carbon adsorption filters and vessels a variety of applications.
What is Type 2 adsorption isotherm?
The reversible Type II isotherm is the normal form of isotherm obtained with a non-porous or macroporous adsorbent. It forms because lateral interactions between adsorbed molecules are strong in comparison to interactions between the adsorbent surface and adsorbate.
Why are isotherms important?
For the adsorption process, an analysis of the isotherm data is important because the isotherms describe equilibrium relationships between adsorbent and adsorbate, usually the ratio between the quantity adsorbed and that remaining in the solution at a fixed temperature at equilibrium.
What is the difference between adsorption capacity and removal efficiency?
qe defines adsorption capacity i.e. unit weight of adsorbate (mg) adsorbent on per gram of adsorbent and is expressed as mg/g. Removal % defines the quantity of adsorbate removed in percentage (%) from a particular volume of adsorbate soluton.
What are the 2 common types of adsorption?
There are two types of adsorption: Physical adsorption and Chemisorption.
How many layers are adsorbed in chemical adsorption?
one layer
Thus, we know that, in the chemical adsorption, one layer is adsorbed. Therefore, the correct answer is option (A).