What is a superheavy element in chemistry?
Definition. Superheavy elements are those elements with a large number of protons in their nucleus. Elements with more than 92 protons are unstable; they decay to lighter nuclei with a characteristic half-life.
How are superheavy elements formed?
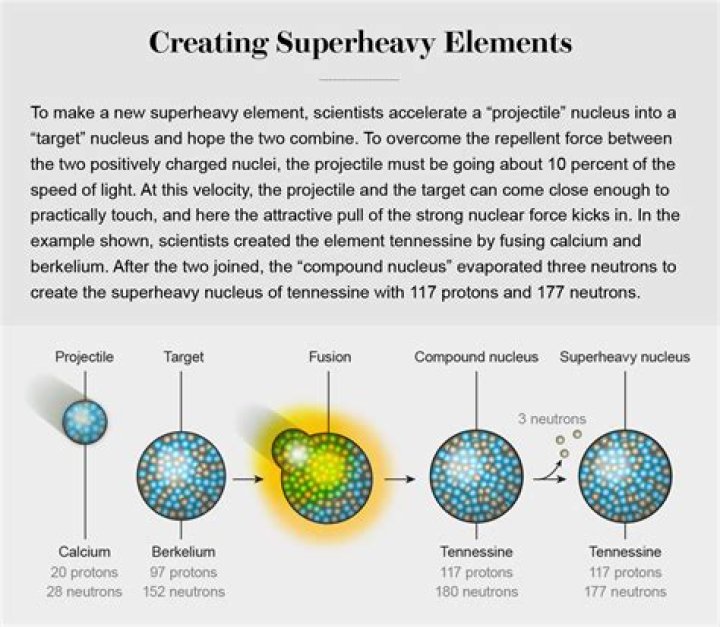
A superheavy atomic nucleus is created in a nuclear reaction that combines two other nuclei of unequal size into one; roughly, the more unequal the two nuclei in terms of mass, the greater the possibility that the two react.
What are superheavy elements used for?
Superheavy elements allow nuclear physicists to explore concepts such as “magic numbers” and the “island of stability”, which help us understand why some nuclei are more stable than others.
How long can superheavy elements live?
Previously known isotopes of lawrencium have fewer neutrons, and are less stable. This novel species, however, has an astonishingly long half-life of 11 hours, making it one of the longest-lived superheavy isotopes known to date.
What is a stable superheavy element?
In nuclear physics, the island of stability is a predicted set of isotopes of superheavy elements that may have considerably longer half-lives than known isotopes of these elements. It is predicted to appear as an “island” in the chart of nuclides, separated from known stable and long-lived primordial radionuclides.
What is class 11 superheavy?
Superheavy elements refer to the element with an atomic number greater than 103 and they are usually radioactive elements. In the periodic table, the known superheavy elements form constituents of the 6d and 7p series.
Why are heavy elements unstable?
The secret is in the very heavy elements made in supernova explosions. Many of the elements heavier than lead have nuclei so large that they are fairly unstable. Due to the instability, over time they eject a neutron or proton, or a neutron in the nucleus decays into a proton and electron.
How did Rutherford synthesize a stable element?
Rutherfordium is created by nuclear bombardment – smashing tiny bits of atoms into other atoms. This is fitting because Ernest Rutherford pioneered the research that first split an atom in 1917. All of the synthetic elements are radioactive and unstable. They do not exist for long because of radioactive decay.
What’s the heaviest element on the periodic table?
uranium
The heaviest naturally stable element is uranium, but over the years physicists have used accelerators to synthesize larger, heavier elements. In 2006, physicists in the United States and Russia created element 118.
Why are superheavy elements radioactive?
Most elements heavier than lead, atomic number 82, are unstable and radioactive. To create these superheavy elements, we smash atoms together to form larger nuclei. But particle accelerator technology, pushed to its limits, has so far only built atoms that decay in as little as a fraction of a microsecond.
What is the largest stable atom?
The periodic table could be considered to end at the largest stable element, lead (atomic number 82). All elements with higher atomic numbers have unstable nuclei, because the assembled protons become too large for the strong nuclear force to hold together.