What is a mixture of atoms and compounds?
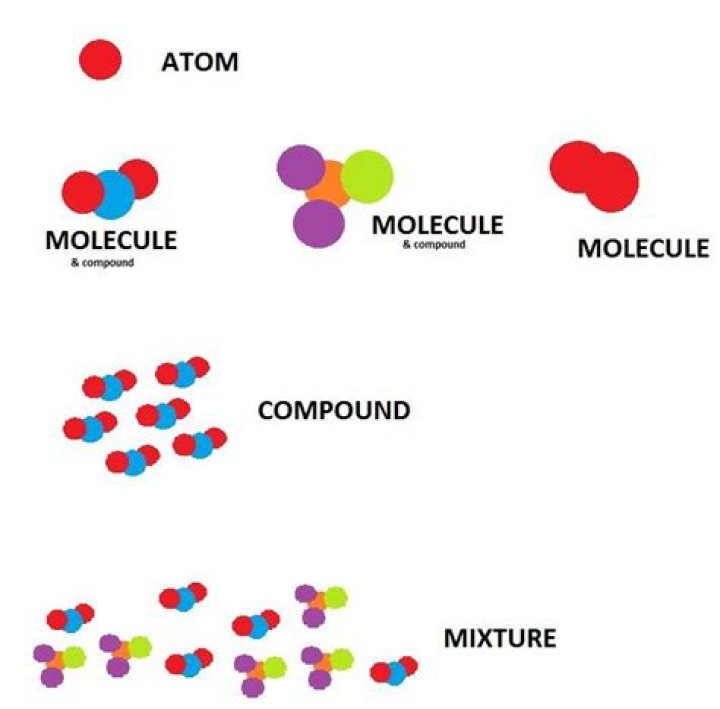
Mixtures are physically combined structures that can be separated into their original components. A chemical substance is composed of one type of atom or molecule. A mixture is composed of different types of atoms or molecules that are not chemically bonded.
What is a mixture of compounds called?
A pure substance consists of a single element or compound. Iron is formed only of iron (Fe) atoms; table salt is formed only by sodium chloride (NaCl) molecules. A mixture, however, is made up of different compounds and/or elements. Homogenous (evenly-distributed) mixtures of two or more chemicals are called solutions.
How are atoms in a compound mixed together?
The elements in compounds are held together by chemical bonds. A chemical bond is a force of attraction between atoms or ions that share or transfer valence electrons. Water is an example of a common chemical compound.
What compounds are made up of atoms?
Compounds are made of two or more atoms of different elements, such as water (H2O) and methane (CH4). Atoms are not drawn to scale. Molecules of compounds have atoms of two or more different elements. For example, water (H2O) has three atoms, two hydrogen (H) atoms and one oxygen (O) atom.
What is difference between compounds and mixture?
Difference between Compound and Mixture Compounds are formed because of chemical bonding between two elements. Mixtures are formed when substances are physically mixed with one another. The composition of compounds is always constant. The composition of mixtures is variable.
Which must be a mixture of substances?
A chemical solution is made of a mixture of two or more substances, the solvent is the substance that does the dissolving and the solute is the substance being dissolved. Learn more about their functions and processes.
What are the examples of compound and mixture?
Difference between Compound and Mixture
| COMPOUNDS | MIXTURES |
|---|---|
| In nature they are homogeneous. | They can be homogeneous as well as heterogeneous in nature. |
| They have a definite melting and boiling point. | They do not have any definite melting or boiling point. |
| Example: Baking soda, salt, etc. | Example: Salt and sand, sugar and water. |
How is a mixture different from a compound?
Compound:a substance that is made up of more than one type of atom bonded together. Mixture: a combination of two or more elements or compounds which have not reacted to bond together; each part in the mixture retains its own properties.
What is compound and mixture with example?
Examples of Mixtures A mixture that contains organic compounds like hydrocarbons forms crude oil. A mixture of water and salt, like seawater. A mixture of gases like nitrogen, oxygen, carbon dioxide, carbon monoxide, etc forms air.
Is a compound always a mixture?
A compound always has the same composition. Mixtures can have different compositions. A compound consists of the atoms of two or more elements that are chemically joined to each other. Since the atoms are always joined in the same ratio, the composition of a compound is always the same.