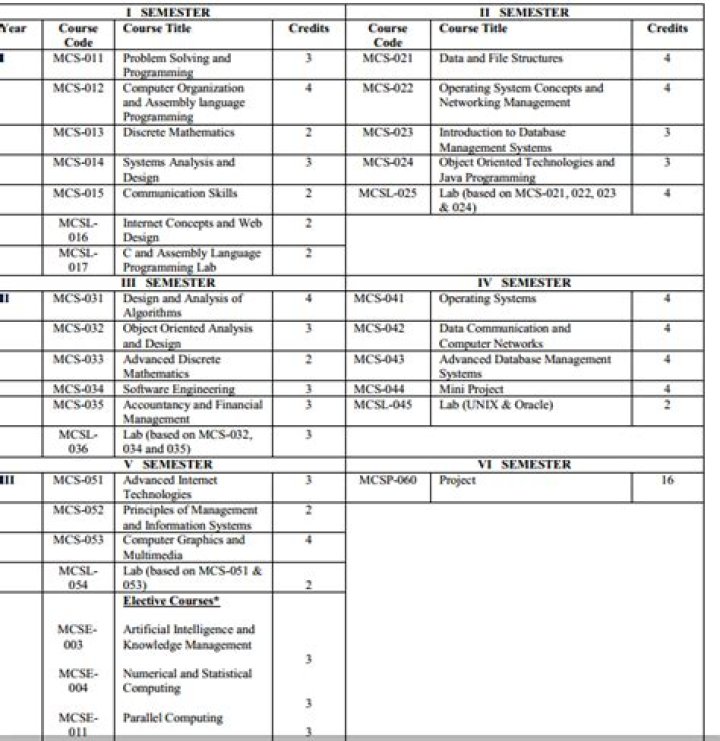
What ionic compound is BaO?
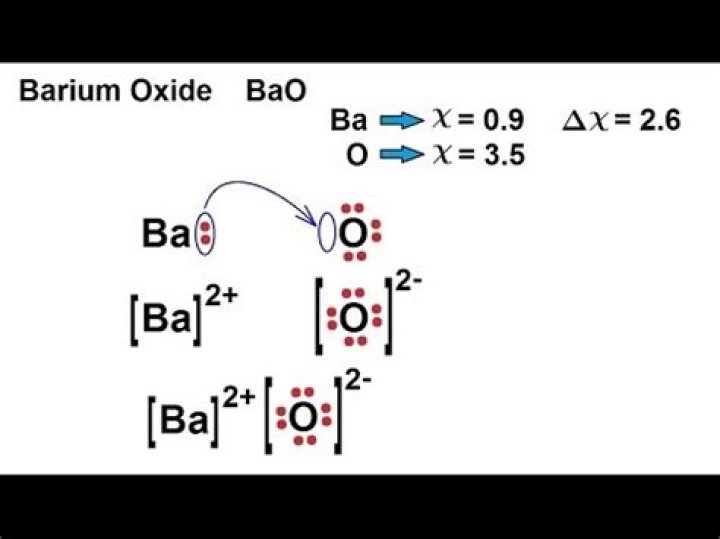
What ionic compound is BaO?
Formula and structure: The barium oxide chemical formula is BaO. The molar mass is 153.33 g/mol. The molecule is formed by one barium cation Ba2+ and one oxide anion O2-. Both ions are bound by one ionic bond.
What is the ionic compound of Ba and O?
Barium oxide is an ionic compound, compromised of a barium metal cation, and an oxygen non-metal anion.
What is the formula for this ionic crystal BaO?
Barium peroxide
| Names | |
|---|---|
| Chemical formula | BaO2 |
| Molar mass | 169.33 g/mol (anhydrous) 313.45 (octahydrate) |
| Appearance | Grey-white crystalline (anhydrous) colorless solid (octahydrate) |
| Odor | odorless |
What is the compound name of bacl2?
Barium chloride
| IUPAC Name | barium(2+);dichloride |
|---|---|
| Alternative Names | Barium chloride Barium dichloride |
| Molecular Formula | BaCl2 |
| Molar Mass | 208.227 g/mol |
| InChI | InChI=1S/Ba.2ClH/h;2*1H/q+2;;/p-2 |
Is BaO a covalent compound?
The most important types of bond are ionic and covalent bonds. Ionic bonds generally occur between metallic elements and nonmetallic elements. Hence, there is a bond between metal and non-metal so the bond formed will be ionic. Therefore, $ BaO $ has an ionic bond.
Is BaO ionic or covalent?
Ionic bonds generally occur between metallic elements and nonmetallic elements. Hence, there is a bond between metal and non-metal so the bond formed will be ionic. Therefore, $ BaO $ has an ionic bond.
How do you find ionic compounds?
To find the formula of an ionic compound, first identify the cation and write down its symbol and charge. Then, identify the anion and write down its symbol and charge. Finally, combine the two ions to form an electrically neutral compound.
What is the name of this compound p2o5?
tricyclo[3.3.1.13,7]tetraphosphoxane 1,3,5,7-tetraoxide
Phosphorus pentoxide/IUPAC ID
What is the scientific name for NaBr?
Sodium bromide (NaBr)
Is Cao an ionic compound?
Oxocalcium
Calcium oxide/IUPAC ID
Is BaO an ionic bond?
In ionic bond, electrons transfer takes place whereas in covalent bonding, sharing of electrons occurs. Ionic bonds generally occur between metallic elements and nonmetallic elements. Hence, there is a bond between metal and non-metal so the bond formed will be ionic. Therefore, $ BaO $ has an ionic bond.