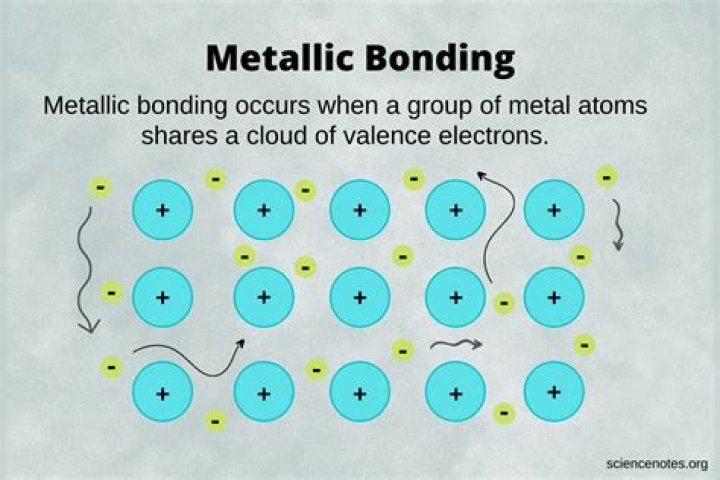
What elements form a metallic bond?
Metallic bonds are formed when the charge is spread over a larger distance as compared to the size of single atoms in solids. Mostly, in the periodic table, left elements form metallic bonds, for example, zinc and copper. Because metals are solid, their atoms are tightly packed in a regular arrangement.
What is an example of a metallic bond?
Like covalent bonds, metallic bonds form between two atoms with similar electronegativity values. Atoms that form metallic bonds are metals and some metalloids. For example, metallic bonds occur in silver, gold, brass, and bronze.
What element is chemically bonded?
When two distinct elements are chemically combined—i.e., chemical bonds form between their atoms—the result is called a chemical compound. Most elements on Earth bond with other elements to form chemical compounds, such as sodium (Na) and Chloride (Cl), which combine to form table salt (NaCl).
What is the bonding in silicon?
covalent bonds
Overview. Each silicon atom has four valence electrons which are shared, forming covalent bonds with the four surrounding Si atoms.
What type of bonding is metallic?
A metallic bond is a type of chemical bond formed between positively charged atoms in which the free electrons are shared among a lattice of cations. In contrast, covalent and ionic bonds form between two discrete atoms. Metallic bonding is the main type of chemical bond that forms between metal atoms.
Is NaCl a metallic bond?
Ionic Bonds: Examples include LiF, NaCl, BeO, CaF2 etc. Covalent Bonds: Examples include hydrogen gas, nitrogen gas, water molecules, diamond, silica etc. Metallic Bonds: Examples include iron, gold, nickel, copper, silver, lead etc.
What is metallic bonding?
What are the 4 types of bonds in chemistry?
There are four types of chemical bonds essential for life to exist: Ionic Bonds, Covalent Bonds, Hydrogen Bonds, and van der Waals interactions.
How many covalent bonds can’s form?
2 single covalent bonds
Sulfur is a nonmetal in group 6A , and therefore has 6 valence electrons. In order to obey the octet rule, it needs to gain 2 electrons . It can do this by forming 2 single covalent bonds.
How does silicon bond to other silicon atoms?
In the silicon crystals that form the backbone of the electronics industry, each silicon atom forms covalent bonds with four other silicon atoms, sharing one of its electrons (and receiving a shared electron in return) from each of the four neighbors.