What element is after tellurium?
What element is after tellurium?
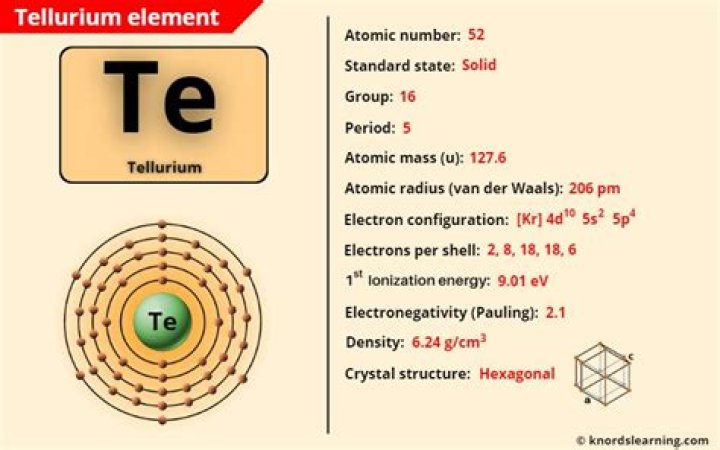
Tellurium is a chemical element with the symbol Te and atomic number 52. It is a brittle, mildly toxic, rare, silver-white metalloid. Tellurium is chemically related to selenium and sulfur, all three of which are chalcogens….Tellurium.
| Hydrogen | Rubidium |
|---|---|
| Strontium | |
| Yttrium | |
| Zirconium | |
| Niobium |
What will the 119 element be?
Element 119 is expected to be a typical alkali metal with a +1 oxidation state. The energetic properties of its valence electron, the 8s electron, suggest that its first ionization potential will be higher than the oxidation potential predicted by simple extrapolation, so that the…
How many electron shells does tellurium have?
Data Zone
| Classification: | Tellurium is a chalcogen and a metalloid |
|---|---|
| Electrons: | 52 |
| Protons: | 52 |
| Neutrons in most abundant isotope: | 78 |
| Electron shells: | 2,8,18,18,6 |
What is tellurium named after?
Word origin: Tellurium comes from the Latin word tellus, which means earth. Discovery: The element was discovered by Muller von Reichenstein in 1782. It was named by German chemist Martin Heinrich Klaproth in 1798.
What is the 38th element?
Strontium – Element information, properties and uses | Periodic Table.
Is element 140 possible?
The current periodic table has proven up for the element with atomic number 118. As of 2020, no elements with an atomic number greater than 118 has successfully incorporated. However, in real-life science, element 140 has yet to be identified.
What are the quantum numbers for tellurium?
Tellurium atoms have 52 electrons and the electronic shell structure is [2, 8, 18, 18, 6] with Atomic Term Symbol (Quantum Numbers) 3P2….Tellurium Atomic and Orbital Properties.
| Atomic Number | 52 |
|---|---|
| Valence Electrons | 5s2 5p4 |
| Oxidation State | -2;-1 1;2;3;4;5;6 |
| Atomic Term Symbol (Quantum Numbers) | 3P2 |
What can tellurium do?
Tellurium has been used to vulcanise rubber, to tint glass and ceramics, in solar cells, in rewritable CDs and DVDs and as a catalyst in oil refining. It can be doped with silver, gold, copper or tin in semiconductor applications. Tellurium has no known biological role.