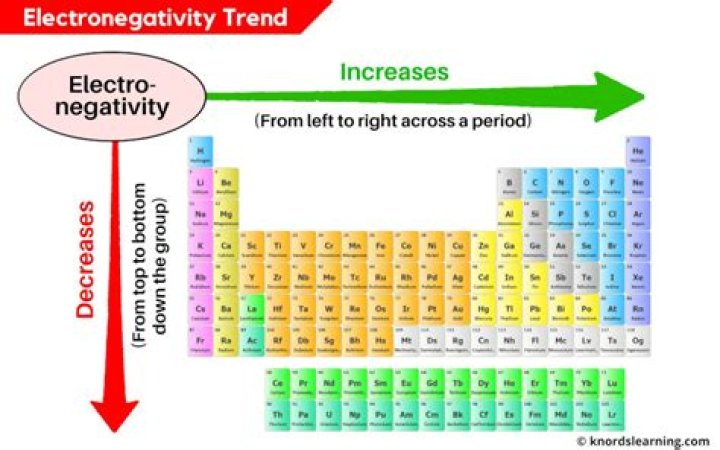
What element has the highest electronegativity of all the elements?
fluorine
Electronegativity increases from bottom to top in groups, and increases from left to right across periods. Thus, fluorine is the most electronegative element, while francium is one of the least electronegative.
What is the most electronegative element?
Fluorine is assinged the oxidation number of -1 because it attracts the electrons in the bond more strongly than the carbon does. Thus, fluorine appears to have an extra electron, -1 oxidation number. F is the most electronegative element on the periodic table.
Where are the elements with the highest electronegativity found?
On the periodic table, electronegativity generally increases as you move from left to right across a period and decreases as you move down a group. As a result, the most electronegative elements are found on the top right of the periodic table, while the least electronegative elements are found on the bottom left.
What is the electronegativity of all elements?
List of Electronegativity Values of the Elements 2
| NUMBER | SYMBOL | ELECTRONEGATIVITY |
|---|---|---|
| 1 | H | 2.20 |
| 2 | He | no data |
| 3 | Li | 0.98 |
| 4 | Be | 1.57 |
Which has higher electronegativity metals or nonmetals?
Nonmetals have much higher electronegativities than metals; of the nonmetals, fluorine is the most electronegative, followed by oxygen, nitrogen, and chlorine. The larger the difference in electronegativity between two atoms, the more polar the bond between them.
Which element has the highest electronegativity in period 3?
In Period 3, sodium with 11 protons is the least electronegative element, and chlorine with 17 protons is the most electronegative element. You might expect argon (with 18 electrons) to be the most electronegative element in Period 3.
What element is the largest?
francium
Thus, helium is the smallest element, and francium is the largest.
Which elements have the highest electronegativity metals or nonmetals?
Why does fluorine have the highest electronegativity?
Fluorine is the most electronegative element because it has 5 electrons in it’s 2P shell. The optimal electron configuration of the 2P orbital contains 6 electrons, so since Fluorine is so close to ideal electron configuration, the electrons are held very tightly to the nucleus.
Do metals have the highest electronegativity?
Following these rules, the non-metals, which are organized on the right side of the periodic table, have higher electronegativity values than the metals. The element with the highest electronegativity is the non-metal fluorine, with an electronegativity value of 4 on the Linus Pauling scale.
How is S electronegative?
2.58Sulfur / Electronegativity