What does thioredoxin reductase do?
Thioredoxin reductase (TR) regulates the intracellular redox environment by reducing thioredoxin (Trx). In anaerobes, recent findings indicate that the Trx redox network is implicated in the global redox regulation of metabolism but also actively participates in protecting cells against O2.
Is thioredoxin reductase an antioxidant?
The thioredoxin (Trx) system, which is composed of NADPH, thioredoxin reductase (TrxR), and thioredoxin, is a key antioxidant system in defense against oxidative stress through its disulfide reductase activity regulating protein dithiol/disulfide balance.
Does thioredoxin reductase contain selenium?
Mammalian thioredoxin reductases (TrxR) are dimers homologous to glutathione reductase with a selenocysteine (SeCys) residue in the conserved C-terminal sequence -Gly-Cys-SeCys-Gly. Thus selenium is required for the catalytic activities of TrxR explaining the essential role of this trace element in cell growth.
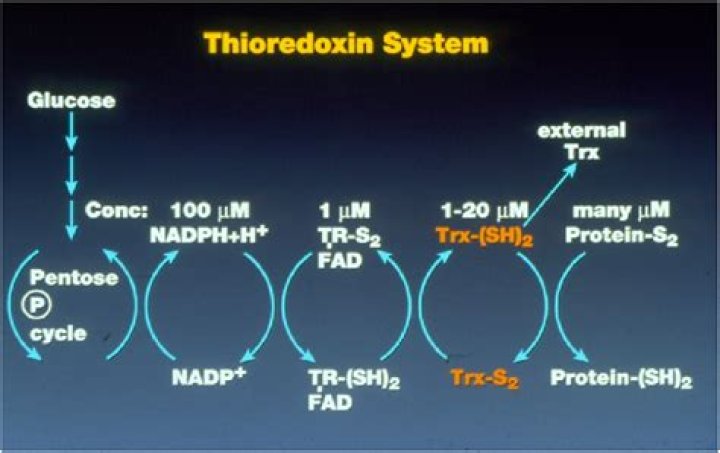
What is thioredoxin system?
The thioredoxin system (TS) consists of an electron donor and two types of antioxidant oxidoreductase proteins: thioredoxin (Trx) and thioredoxin reductase (TrxR) and NADPH as the electron donor. Trx was first identified as a hydrogen donor for ribonucleotide reductase in Escherichia coli [1.
What is thioredoxin in plants?
Thioredoxins are small proteins catalyzing thiol-disulfide interchange and are involved in the regulation of the redox environment of the cell. In plants, the thioredoxin system is particularly complex since at least 20 thioredoxin isoforms are found in the plant model Arabidopsis thaliana.
What is the function of glutathione peroxidase?
Glutathione peroxidase is an antioxidant enzyme class with the capacity to scavenge free radicals. This is in turn helps to prevent lipid peroxidation and maintain intracellular homeostasis as well as redox balance [71].
What does Glutaredoxin c1 do?
Glutaredoxins function as electron carriers in the glutathione-dependent synthesis of deoxyribonucleotides by the enzyme ribonucleotide reductase. Moreover, GRX act in antioxidant defense by reducing dehydroascorbate, peroxiredoxins, and methionine sulfoxide reductase.
What is regulated by thioredoxin?
Thioredoxin regulates the activity of various substrates, including those that function to counteract cellular oxidative stress. These include the peroxiredoxins, methionine sulfoxide reductase A and specific transcription factors. Several of these transcription factors are in turn redox regulated.
What is an essential part of the glutathione peroxidase enzyme system?
Selenium is part of the glutathione peroxidase system. b.
What is the difference between glutathione and glutathione peroxidase?
Glutathione, which is involved in the transport of amino acids, acts as a coenzyme for enzymes and it protects against oxygen radicals and toxic compounds [5]. Se- dependent GSH peroxidase is capable of utilizing hydrogen peroxide (H2O2) and a variety of organic hydroperoxides as substrates.
What does Glutaredoxin do in plants?
Glutaredoxins (GRXs) are glutathione-dependent disulfide oxidoreductases that are well documented to be involved in oxidative stress responses in plants (reviewed in Rouhier et al., 2008). GRXs allow for redox regulation of protein activity by reversibly glutathionylating or reducing disulfide bridges in their targets.
Is Glutaredoxin an enzyme?
Oxidation and Reduction of Biological Material Glutaredoxins (Grx) are small intracellular redox enzymes. Like thioredoxins, they contain in the reduced state two redox-active thiols that form a disulfide bond on oxidation. Unlike Trx, oxidized glutathione is reduced by glutathione reductase [290].