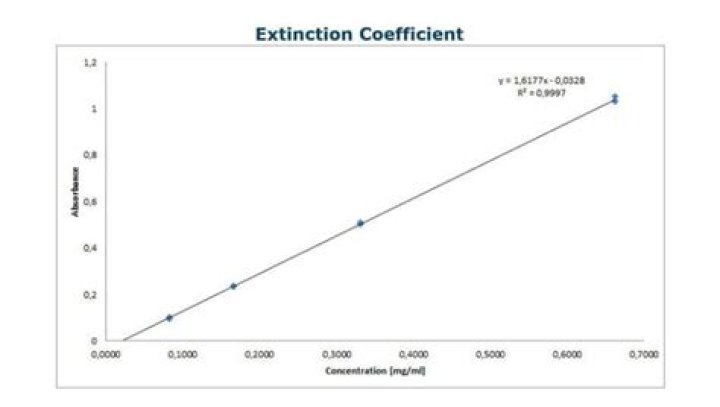
What does the extinction coefficient measure?
Extinction coefficient, a measure of how strongly a substance absorbs light at a specific wavelength, is the intrinsic property of a protein depending on its composition and structure. Hence, to precisely determine protein concentration, it is fundamental to accurately determine extinction coefficient.
What is atmospheric extinction coefficient?
The atmospheric extinction coefficient (bext), which is the sum of scattering and absorption by particles and gases, is a measure of the alteration of radiant energy as it passes through the atmosphere.
What factors affect extinction coefficient?
ε is the molar extinction coefficient….The three factors include:
- The amount of light absorbed by the substance for a specific wavelength.
- The distance that the light travels through the solution.
- The concentration of the absorbing solution per unit volume.
What is the difference between absorption coefficient and extinction coefficient?
The extinction coefficient is a measure of the damping of the electromagnetic wave as it passes into a medium. The absorption coefficient, a, allows us to measure how much light is absorbed and is related to k by a=(4*pi*k)/lambda. It essentially the reciprocal of how far the light travels into the solid.
Does extinction coefficient depend on wavelength?
The refractive index (n) and extinction coefficient (k) are related to the interaction between a material and incident light and are associated with refraction and absorption, respectively. Both refractive index (n) and extinction coefficient (k) depend on the wavelength.
Does molar extinction coefficient change with wavelength?
The Molar Absorptivity Constant is specific for every single solution, and at every wavelength. When you are taking an absorbance spectrum, and measuring the absorbance at different wavelengths, this is the only factor that is changing, as the concentration of the solution remains the same, and so does the pathlength.