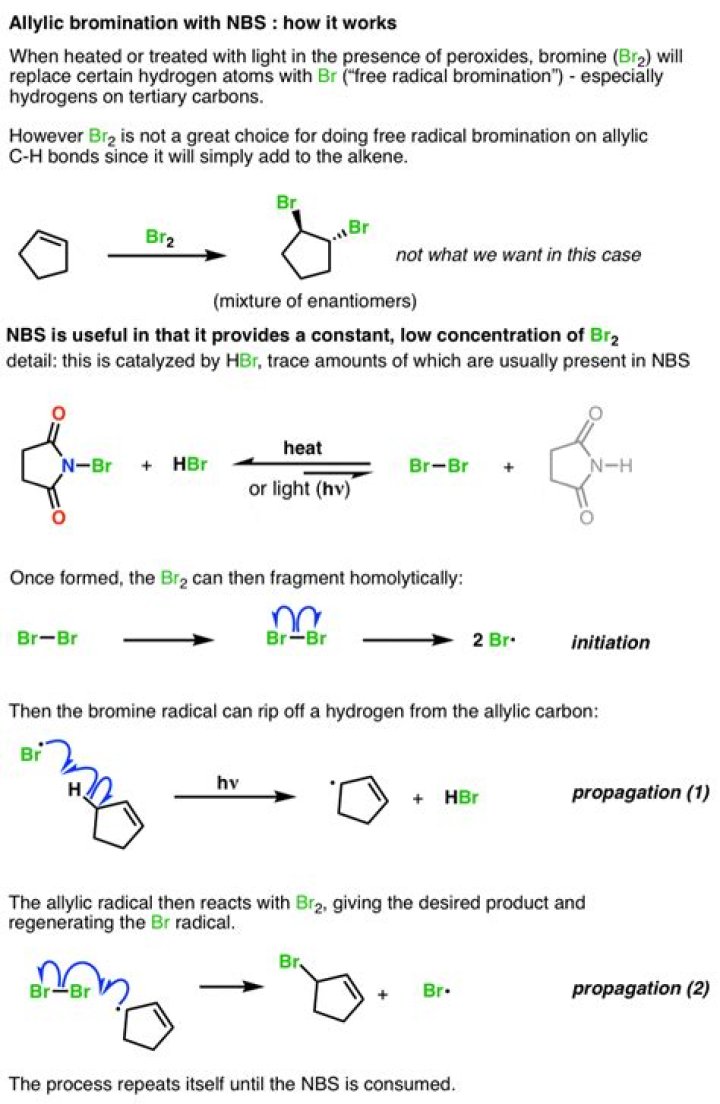
What does NBS do in reaction?
N-Bromosuccinimide (NBS) is a brominating and oxidizing agent that is used as source for bromine in radical reactions (for example: allylic brominations) and various electrophilic additions.
What does NBS do to an alkene?
Alkenes react with NBS in dry CCl4 under reflux conditions to give allyl bromide. The reaction is initiated by light or peroxide. Although a number of reagents are available for bromination of allylic C-H bond of alkenes, NBS is most commonly used. The reaction is called Wohl-Zigler bromination.
What does NBS stand for in Chem?
N-Bromosuccinimide or NBS is a chemical reagent used in radical substitution, electrophilic addition, and electrophilic substitution reactions in organic chemistry.
Is NBS an electrophile?
In contrast, NBS (N-bromo succinimide) is a gleaming white crystalline solid and easy as pie to work with. But don’t be deceived. It will do many of the same reactions as bromine – attached to the electron-withdrawing nitrogen of succinimide, the bromine has a partial positive charge and is therefore electrophilic.
What does toluene do in a reaction?
Chemical properties Toluene reacts as a normal aromatic hydrocarbon in electrophilic aromatic substitution. Because the methyl group has greater electron-releasing properties than a hydrogen atom in the same position, toluene is more reactive than benzene toward electrophiles.
What type of reaction is the nitration of toluene?
electrophilic aromatic substitution reaction
Hint: Before answering this question, we should know that nitration of Toluene involves electrophilic aromatic substitution reaction. It involves addition of nitrate in ortho and meta position of an aromatic compound.
What is the major product formed when Methylenecyclohexane reacts with NBS?
1-(bromomethyl)cyclohexene.
Can be prepared from toluene by chlorination with?
D. NaOCl. Hint: In methods of preparation of aromatic halides we have studied that benzyl chloride can be formed from toluene by chlorination with $C{l_2}$ because chlorine can give free radical in presence of sunlight. This reaction of chlorination is a free radical substitution reaction.
When toluene is reacted with bromine in presence of UV light HN The product formed will be?
benzyl bromide
Toluene reacts with bromine in the presence of light to give benzyl bromide, while in the presence of FeBr3, it gives p−bromotoluene.