What does limestone do in scrubbers?
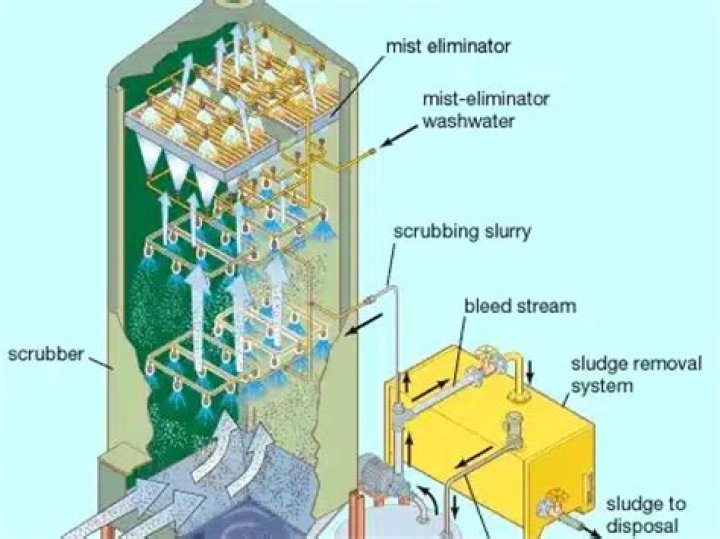
A system for removal of SO2 from pollution sources by injecting calcined limestone into the exhaust gas system. In a wet scrubber (the most common type), the absorbent, a mixture of water and limestone, is sprayed inside a treatment area onto the flue gas, which rises from the bottom of the absorber area.
What chemical is formed when limestone calcium carbonate scrubbers are used to control sulfur dioxide?
If a dry scrubber is used, the flue gas comes into contact with pulverized limestone – which is mainly calcium carbonate. The chemical reaction between the calcium carbonate and the sulfur dioxide yield calcium sulfite. This calcium sulfite either falls out of the gas stream or is removed with other particulates.
Why lime scrubbing is preferred as compare to limestone scrubbing for flue gas desulfurization?
Lime is more reactive than limestone, and requires less capital equipment. SO2 removal efficiencies using lime scrubbers range from 95 to 99 percent at electric generating plants. There are two main methods for cleaning flue gases from coal combustion at electric generating stations: dry scrubbing and wet scrubbing.
How does limestone remove sulfur dioxide?
Limestone is a naturally occurring calcium carbonate rock which can react with sulfur dioxide. Limestone can therefore be used for desulfurization of flue gases. The reaction of sulfur di- oxide with dry limestone requires a very high temperature such as that available within the actual boiler furnace.
What is a disadvantage of limestone scrubbers?
Other drawbacks of wet-limestone scrubbing are large up-front capital costs, large equipment size and substantial predictive and corrective maintenance requirements. Substituting hydrated lime as the reagent reduces equipment size and costs, but increases reagent costs and material handling issues.
How is lime manufactured from limestone?
The basic processes in the production of lime are: (1) quarrying raw limestone; (2) preparing limestone for the kilns by crushing and sizing; (3) calcining limestone; (4) processing the lime further by hydrating; and (5) miscellaneous transfer, storage, and handling operations.
How does limestone react with sulfur dioxide?
At high temperature, the limestone or dolomite rock is usually calcined immediately, and, in the presence of excess oxygen, sulfur dioxide combines with calcium oxide to form calcium sulfate.
What is limestone slurry?
Lime slurry is a suspension of calcium hydroxide in water. The product is a user-friendly, cost-effective alkali. Markets. Lime slurry is a free-flowing product that is used for a variety of industrial, municipal and environmental applications.
What is wet lime?
Wet lime systems are well established in some industries such as Power Generation & Waste to Energy. This process is based on the large degree of contact between the circulating slurry and the flue gases. This slurry circulates in the absorber tower (or scrubber) to neutralize the condensed flue gas pollutant.
What percentage of limestone is calcium carbonate?
Composition of Limestone Limestone is by definition a rock that contains at least 50% calcium carbonate in the form of calcite by weight. All limestones contain at least a few percent other materials.
What is decomposition of limestone?
Limestone is calcium carbonate (CaCO3) which when heated to above 840°C decomposes into calcium oxide (CaO) while releasing carbon dioxide (CO2) according to the equation: CaCO3(s) → CaO(s) + CO2(g). it is a decomposition reaction.