What causes high specific heat in water?
Water has a higher specific heat capacity because of the strength of the hydrogen bonds. It requires a significant of energy to separate these bonds.
What is high specific heat of water?
Specific Heat of Water Water has a bizarrely high specific heat (in general, it’s the highest of all liquids) due to hydrogen bonding between molecules. The specific heat is the amount of heat needed to raise a gram of water by one degree Celsius.
What does it mean when the specific heat is high?
Specific heat is how much heat energy is needed raise the temperature of a substance. Water has a very high specific heat. That means it needs to absorb a lot of energy before its temperature changes. This means that their temperatures change more quickly.
What causes specific heat?
This quantity is known as the specific heat capacity (or simply, the specific heat), which is the heat capacity per unit mass of a material. Experiments show that the transferred heat depends on three factors: (1) The change in temperature, (2) the mass of the system, and (3) the substance and phase of the substance.
Why does water have high latent heat of vaporization?
Thus a liquid can flow, unlike a solid. The energy required to completely separate the molecules, moving from liquid to gas, is much greater that if you were just to reduce their separation, solid to liquid. Hence the reason why the latent heat of vaporization is greater that the latent heat of fusion.
What is the specific heat of liquid water?
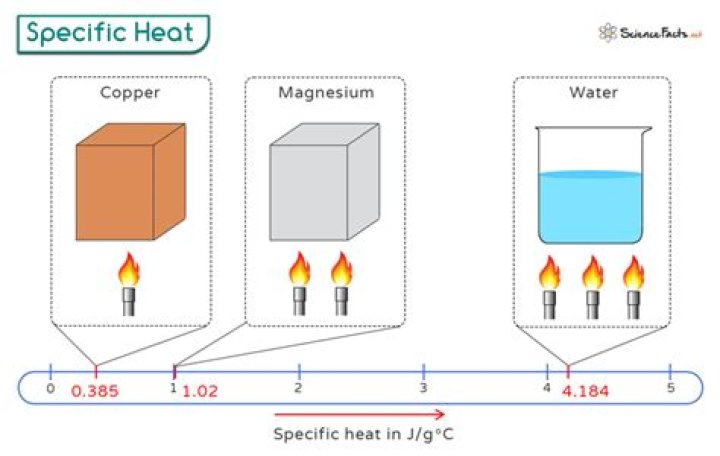
about 4184 J⋅kg
Liquid water has one of the highest specific heat capacities among common substances, about 4184 J⋅kg−1⋅K−1 at 20 °C; but that of ice, just below 0 °C, is only 2093 J⋅kg−1⋅K−1.
Why does water have a higher specific heat than ethanol?
So an obvious question is, why is the specific heat of water so much higher than that of these alcohols? Because there are more hydrogen bonds forming attractions between water molecules, it takes more energy to overcome those interactions and raise the kinetic energy of the water molecules.
How do you find the specific heat of water?
Calculate specific heat as c = Q / (mΔT) . In our example, it will be equal to c = -63,000 J / (5 kg * -3 K) = 4,200 J/(kg·K) . This is the typical heat capacity of water.
What does high latent heat of vaporization mean?
Likewise, the high latent heat of vaporization (see below), indicates that when water vapor (derived from evaporation of water at the ocean’s surface driven by solar energy receipt at low latitudes) condenses into liquid droplets at high elevations or high latitude, the latent heat is released into the environment.
How does water absorb heat?
Water absorbs heat by vibrating and rotating. The vibrations can be the stretching or bending of the chemical bonds between hydrogen and oxygen. Answer 4: If the water is cooler than the air, then heat energy will “flow” from the air into the water until the temperatures are equal.