What atom has 16 protons 17 neutrons?
The sulfur atom has a mass of 32⋅amu .
Which of the following elements has 17 electrons and 17 protons?
chlorine
The atomic number of chlorine is 17. So, it has 17 electrons and 17 protons.
What is the element with 16 protons?
Sulfur
Sulfur is a chemical element. Its official symbol is S and its atomic number is 16, which means that each sulfur atom has 16 protons in its nucleus.
How many protons does magnesium have?
12
Magnesium/Atomic number
The most common and stable type of magnesium atom found in nature has 12 protons, 12 neutrons, and 12 electrons (which have a negative charge). Atoms of the same element with different neutron counts are known as isotopes.
What isotope has 16 protons and 16 neutrons?
So, S-32 has 16 protons and 16 neutrons. The number of electrons is equal to the number of protons for all elements (they have no net charge). So S-32 also has 16 electrons.
Which of the following is true about element with atomic number 18?
its electronegativity is very high. element with atomic number 18 noble gas (Ar).
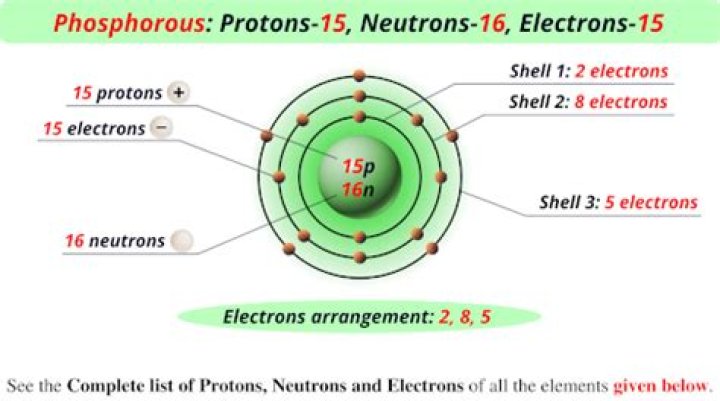
Which of the following is true about an element that has atomic number 15?
The element with atomic number $ 15 $ is phosphorus. Thus, phosphorus has similar properties to nitrogen, arsenic, antimony, and bismuth. In the given options, nitrogen is present.
What atom has 17 protons and 18 neutrons and 17 electrons?
chlorine atom
A chlorine atom has 17 protons, 18 neutrons, and 17 electrons.
What is the name of the element that has 17 protons?
Chlorine-37
| General | |
|---|---|
| Symbol | 37Cl |
| Names | chlorine-37, Cl-37 |
| Protons | 17 |
| Neutrons | 20 |
What is the chemical symbol of magnesium?
Mg
Magnesium/Symbol