What are the signs of Delta H and Delta S for a reaction that is spontaneous under all conditions?
A spontaneous reaction will always occur when Delta H is negative and Delta S is positive, and a reaction will always be non-spontaneous when Delta H is positive and Delta S is negative.
How do you know if its spontaneous or Nonspontaneous?
A mathematical combination of enthalpy change and entropy change allows the change in free energy to be calculated. A reaction with a negative value for ΔG releases free energy and is thus spontaneous. A reaction with a positive ΔG is nonspontaneous and will not favor the products.
What are the signs for ΔH and ΔS for a reaction that is product favored at low temperature but reactants favored at high temperature?
Standard Gibbs Free-Energy Change
| sign of ΔH° | sign of ΔS° | Meaning |
|---|---|---|
| + | + | The sign of ΔG° is temperature dependent. The reaction will be product-favored at relatively high temperatures. |
| _ | _ | The sign of ΔG° is temperature dependent. The reaction will be product-favored at relatively low temperatures. |
Under what condition will a reaction be spontaneous if both Delta H and Delta S are negative?
If ΔH and ΔS are both negative, ΔG will only be negative below a certain threshold temperature and we say that the reaction is only spontaneous at ‘low temperatures.
What is the relation between Delta G and Delta H?
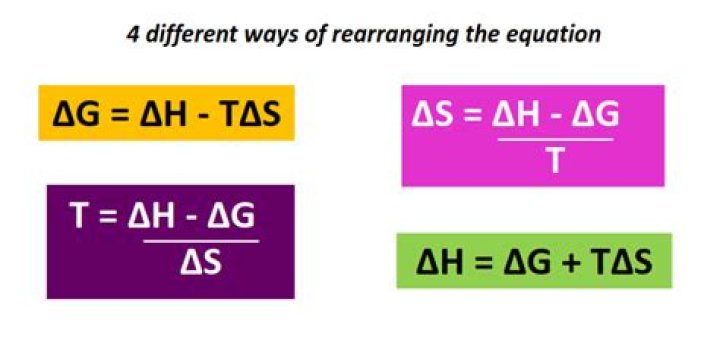
ΔH=ΔG+TΔS.
What is Delta G and Delta S?
∆G is the change in free energy. Keq is the equilibrium constant (remember Keq = [products]/[reactants] ∆H is the change in enthalpy from reactants to products. ∆S is the change in entropy (disorder) from reactants to products. R is the gas constant (always positive)
How do you know if a reaction is spontaneous by Delta G?
When ΔH is negative and ΔS is positive, the sign of ΔG will always be negative, and the reaction will be spontaneous at all temperatures. This corresponds to both driving forces being in favor of product formation.
What are the signs of H and S for the reaction which is spontaneous at low temperature and non-spontaneous at very high temperature?
Reactions with a positive ∆H and negative ∆S are non-spontaneous at all temperatures. Reactions with a negative ∆H and negative ∆S are spontaneous at low temperatures, while reactions with a positive ∆H and positive ∆S are spontaneous at high temperatures.
How do you know if a reaction is endothermic or exothermic delta H?
So, if a reaction releases more energy than it absorbs, the reaction is exothermic and enthalpy will be negative. Think of this as an amount of heat leaving (or being subtracted from) the reaction. If a reaction absorbs or uses more energy than it releases, the reaction is endothermic, and enthalpy will be positive.
When Delta H is positive and Delta S is negative?
When ΔH is positive and ΔS is negative, the sign of ΔG will always be positive, and the reaction can never be spontaneous. This corresponds to both driving forces working against product formation. When one driving force favors the reaction, but the other does not, it is the temperature that determines the sign of ΔG.
Under what conditions will this reaction be spontaneous?
When ΔS > 0 and ΔH < 0, the process is always spontaneous as written. When ΔS < 0 and ΔH > 0, the process is never spontaneous, but the reverse process is always spontaneous. When ΔS > 0 and ΔH > 0, the process will be spontaneous at high temperatures and non-spontaneous at low temperatures.