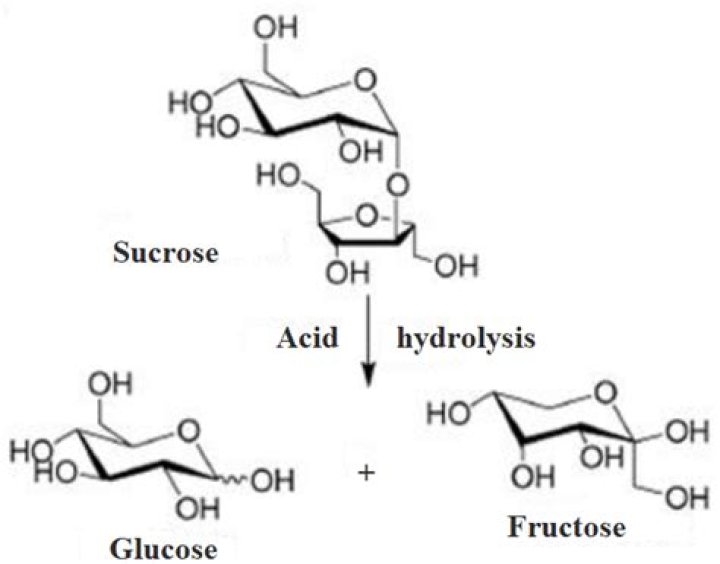
What are the products of acid hydrolysis of sucrose?
Thus, the acidic hydrolysis of sucrose is carried out in the acidic medium such as hydrochloric acid. The products formed are glucose and fructose.
What happens during hydrolysis of sucrose?
Hydrolysis of sucrose yields glucose and fructose. Invertase is a sucrase used industrially for the hydrolysis of sucrose to so-called invert sugar.
What is the equation of hydrolysis of sucrose?
The rate equation for the hydrolysis of sucrose to fructose and glucose, C12H22O11(aq)+H2O(l)→2C6H12O6(aq), C 12 H 22 O 11 ( a q ) + H 2 O ( l ) → 2 C 6 H 12 O 6 ( a q ) , is [sucrose]/t = k[C12H22O11] k [ C 12 H 22 O 11 ] .
What is the order of the acidic hydrolysis of sucrose?
second order reaction. unimolecular raction. It is an example of pseudo unimolecular reaction where molecularity ≥ 2 but order of reaction is one .
What is hydrolysis of sucrose called?
Hydrolysis of sucrose to give glucose and fructose is called inversion. because of the angle of the specific rotation of the plane-polarized light changes from a positive to a negative value i.
What is the product of complete hydrolysis of sucrose?
fructose
The hydrolysis of sucrose generates an equimolar mixture of fructose and glucose, commercially known as invert sugar.
How does invertase break down sucrose?
This enzyme involve in the transport of sucrose by balancing the sucrose concentration of phloem loading and unloading [11]. Invertase break down the sucrose into glucose and fructose as a result increase in osmotic pressure of cells, so this play role in cell elongation and growth process [12].
How does hydrolysis of sucrose change the measurement?
On hydrolysis, the enthalpy decreases by 14.4 kJ/mol of sucrose at 310 K, and the heat capacity, Cp, increases by 61 J mol-1 K-1 of sucrose in the solution. Cp of the solution increases with temperature less rapidly before sucrose hydrolysis than after it.
Why is hydrolysis of sucrose a first order reaction?
In the reaction, water is present in excess and its concentration is taken to be constant, thus the reaction becomes independent of [H2O] and is pseudo first order.
What type of a reaction is hydrolysis of sucrose justify?
It is an example of pseudo unimolecular reaction where molecularity ≥2 but order of reaction is one.
Why is hydrolysis of sucrose inversion?
When sucrose is hydrolyzed it forms a 1:1 mixture of glucose and fructose. It is called invert sugar because the angle of the specific rotation of the plain polarized light changes from a positive to a negative value due to the presence of the optical isomers of the mixture of glucose and fructose sugars.
Is hydrolysis of sucrose a first order reaction?
The hydrolysis of sucrose into glucose and fructose in acidic water is a first-order reaction.