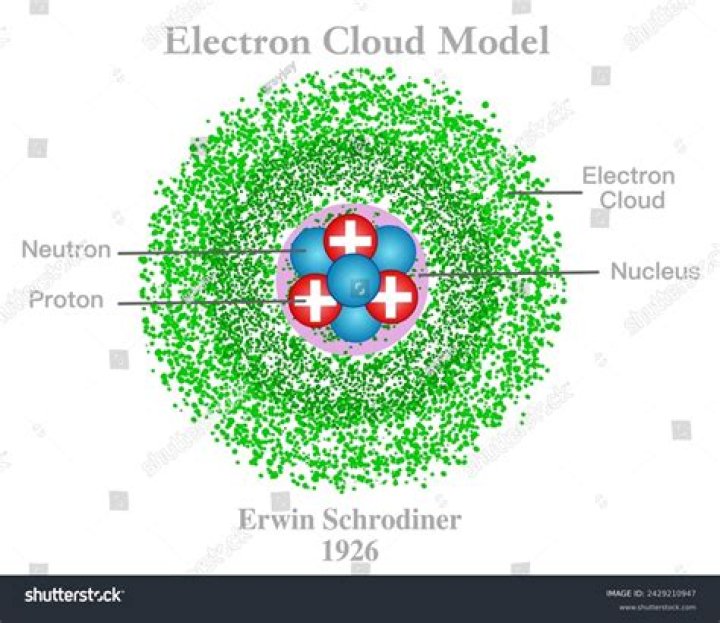
What are the parts of the electron cloud model?
The current model of the atom is called the Electron Cloud Model. In this model, the atom is made of three basic parts, protons, neutrons, and electrons. The protons are positively-charged particles, and the neutrons are particles that do not have an electrical charge.
What is a characteristic of an electron cloud?
An electron cloud represents the area around an atom’s nucleus where electrons are most likely to be found. The cloud is darkest at the nucleus and lighter farther away, representing that electrons are more likely to be found closer to the nucleus than away from it.
What was the electron cloud model?
Austrian physicist Erwin Schrödinger (1887-1961) developed an “Electron Cloud Model” in 1926. It consisted of a dense nucleus surrounded by a cloud of electrons at various levels in orbitals. Schrödinger and Werner Heisenburg (1901-1976) mathematically determined regions in which electrons would be most likely found.
What makes the electron cloud model different from the other atomic models?
Based on quantum theory, which states that all matter has properties associated with a wave function, the Electron Cloud Model differs from the Bohr Model in that it does not define the exact path of an electron.
What is an electron cloud made up of?
Electron cloud: A group of electrons circulating around a nucleus or a molecule. Usually refers to the valence electrons.
Who made the electron cloud model?
Erwin Schrodinger
One such example is the Electron Cloud Model proposed by Erwin Schrodinger. Thanks to this model, electrons were no longer depicted as particles moving around a central nucleus in a fixed orbit. Instead, Schrodinger proposed a model whereby scientists could only make educated guesses as to the positions of electrons.
What are the characteristics of electrons?
The electron is a low-mass, negatively charged particle. As such, it can easily be deflected by passing close to other electrons or the positive nucleus of an atom. The electron was the first fundamental particle that was discovered.
How are orbit and electron cloud models of the atom different?
Explanation: Bohr’s model treats electron energy levels as clearly defined orbital paths around the nucleus ( ike planets orbit the Sun). The cloud model treats the energy levels as probability clouds, i.e. regions in which electrons are likely to be found.
How is the electron cloud model different to the atomic models proposed by Bohr and Rutherford?
Bohr thought that electrons orbited the nucleus in quantised orbits. Bohr built upon Rutherford’s model of the atom. In Rutherford’s model most of the atom’s mass is concentrated into the centre (what we now call the nucleus) and electrons surround the positive mass in something like a cloud.
Which model is the most scientifically accurate model that we have of the atom currently?
The electron cloud model is currently the most sophisticated and widely accepted model of the atom. It retains the concept of the nucleus from Bohr and Rutherford’s models, but introduces a different definition of the motion of electrons around the nucleus.