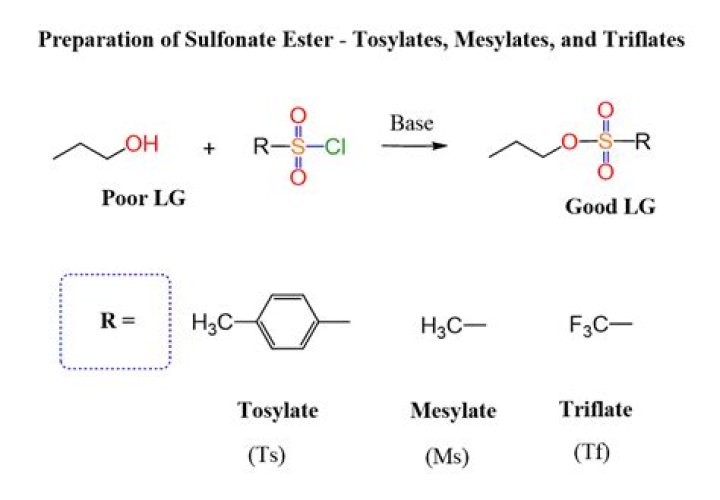
What are Mesylates and Tosylates?
It’s relatively straightforward actually. We use “mesyl chloride” (MsCl) or “tosyl chloride” (TsCl), and the neutral alcohol performs a substitution reaction on sulfur, leading to formation of O-S and breakage of S-Cl. Then, deprotonation of the charged alcohol leads to the neutral mesylate or tosylate.
What does TsCl do in a reaction?
Tosyl chloride (TsCl) is usually used as an activating group for primary alcohols. Due to its relatively large volume and the lower reactivity of secondary and tertiary alcohols, it usually doesn’t come into them, being selective to primary alcohols in most of the cases.
How is mesylate prepared?
Preparation. Mesylates are generally prepared by treating an alcohol and methanesulfonyl chloride in the presence of a base, such as triethylamine.
What is TsCl in organic chemistry?
This white, malodorous solid is a reagent widely used in organic synthesis. Abbreviated TsCl or TosCl, it is a derivative of toluene and contains a sulfonyl chloride (−SO2Cl) functional group.
Is mesylate or triflate better?
No. They are the best leaving groups, but there are other good leaving groups. Triflate, tosylate, and mesylate ions are excellent leaving groups, because the sulfonate ions can stabilize the negative charge via resonance. These ions are weak bases because they are the conjugate bases of very strong sulfonic acids.
Is mesylate a good leaving group?
Chlorides, bromides, and tosylate / mesylate groups are excellent leaving groups in nucleophilic substitution reactions, due to resonance delocalization of the developing negative charge on the leaving oxygen.
What is the common role of a tosylate group?
Tosyl (Ts) group is commonly used as a protecting group for amines in organic synthesis.
What does OTs stand for in organic chemistry?
OTs in organic chemistry stands for tosylate. It is a functional group which is formed by the loss of the hydrogen of OH in para-toluenesulfonic…
Is tosylate or mesylate a better leaving group?
, mesylate and tosylate are better leaving groups than alcohols are. Usually, the best leaving groups are very weak bases. Methanesulfonic acid and p-toluenesulfonic acid are extremely strong acids, Their conjugate bases are extremely weak, so the conjugate bases are excellent leaving groups.
Is oxygen a good leaving group?
Oxygen makes a poor leaving group in substitution and elimination reactions. That is, when left in its initial form. The key to kicking out an oxygen is to ‘bribe it’ by turning it into a better leaving group.