What are 5 examples of polyatomic ions?
Examples of Common Polyatomic Ions
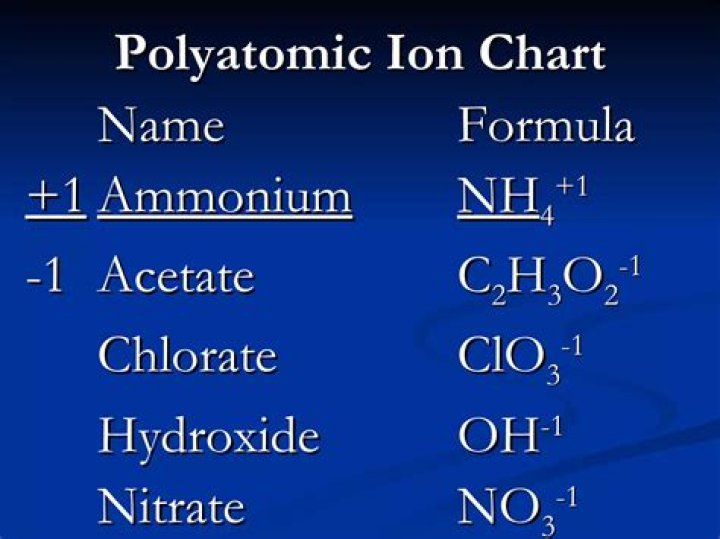
- Acetate – C2H3O2–
- Bicarbonate (or hydrogen carbonate) – HCO3–
- Bisulfate (or hydrogen sulfate) – HSO4–
- Hypochlorite – ClO–
- Chlorate – ClO3–
- Chlorite – ClO2–
- Cyanide – CN-
- Hydroxide – OH-
What ions are polyatomic?
Polyatomic ions are ions which consist of more than one atom. For example, nitrate ion, NO3-, contains one nitrogen atom and three oxygen atoms. The atoms in a polyatomic ion are usually covalently bonded to one another, and therefore stay together as a single, charged unit.
What are the 13 polyatomic ions?
Terms in this set (13)
- Ammonium. NH4+
- Acetate. C2H3O2-
- Nitrate. NO3-
- Cyanide. CN-
- Hydroxide. OH-
- Permanganate. MnO4-
- Chlorate. ClO3-
- Perchlorate. ClO4-
What are 3 examples of polyatomic ions?
Well-known examples of such polyatomic ions are the sulfate ion (SO42–), the hydroxide ion (OH–), the hydronium ion (H3O+), and the ammonium ion (NH4+).
What do you mean by polyatomic ion give one example?
A polyatomic ion, also known as a molecular ion, is a charged chemical species(ion) composed of two or more atoms covalently bonded or of a metal complex that can be considered to be acting as a single unit. Eg: SO42−, CO32−.
What is the polyatomic ion in Mg OH 2?
polyatomic ions and compounds names
| A | B |
|---|---|
| Magnesium hydroxide | Mg(OH)2 |
| Aluminum chlorate | Al(ClO3)3 |
| Sodium nitrite | NaNO2 |
| Lithium sulfite | Li2 SO3 |
What are the polyatomic ions give two examples?
The prefix poly- means “many,” in Greek, but even ions of two atoms are commonly referred to as polyatomic. Examples: Carbonate (CO32-), sulphate (SO42-), nitrate – NO3- and hydroxide ions OH- are all polyatomic ions.
How many polyatomic ions are there?
Polyatomic Formulas – Polyatomic ions are made from more that one atom. This group of atoms act together as one unit with a single charge. Each of the polyatomic ions have a unique name….
| The 9 polyatomic ions to know and write on your notecard: | ||
|---|---|---|
| Name | Charge | Formula |
| Phosphate | 3- | PO43- |
| Hydronium | 1+ | H3O+ |
| Ammonium | 1+ | NH4+ |
What are polyatomic ions give examples Class 9 answer?
Some examples are: NO−3, this is nitrate ion and it is regarded as a polyatomic ion because there is one nitrogen and three oxygen atoms and it has a negative charge. SO2−4, this is sulfate ion and it is regarded as a polyatomic ion because there is one sulfur and four oxygen atoms and it has a -2 charge.
What is the difference between molecules 2O and O2?
The difference between O2 and 2O is that O2 is a molecule consisting of 2 atoms of oxygen while 2O consists of two separate oxygen atoms.