What are 5 examples of combustion reactions?
Examples of Combustion Reactions
- Combustion of methane. CH4(g) + 2 O2(g) → CO2(g) + 2 H2O(g)
- Burning of naphthalene.
- Combustion of ethane.
- Combustion of butane (commonly found in lighters)
- Combustion of methanol (also known as wood alcohol)
- Combustion of propane (used in gas grills, fireplaces, and some cookstoves)
What are the products of a combustion of a hydrocarbon?
Complete combustion of hydrocarbons (without impurities) in the presence of enough oxygen produces water vapor and carbon dioxide.
Is hydrocarbon a reactant in a combustion reaction?
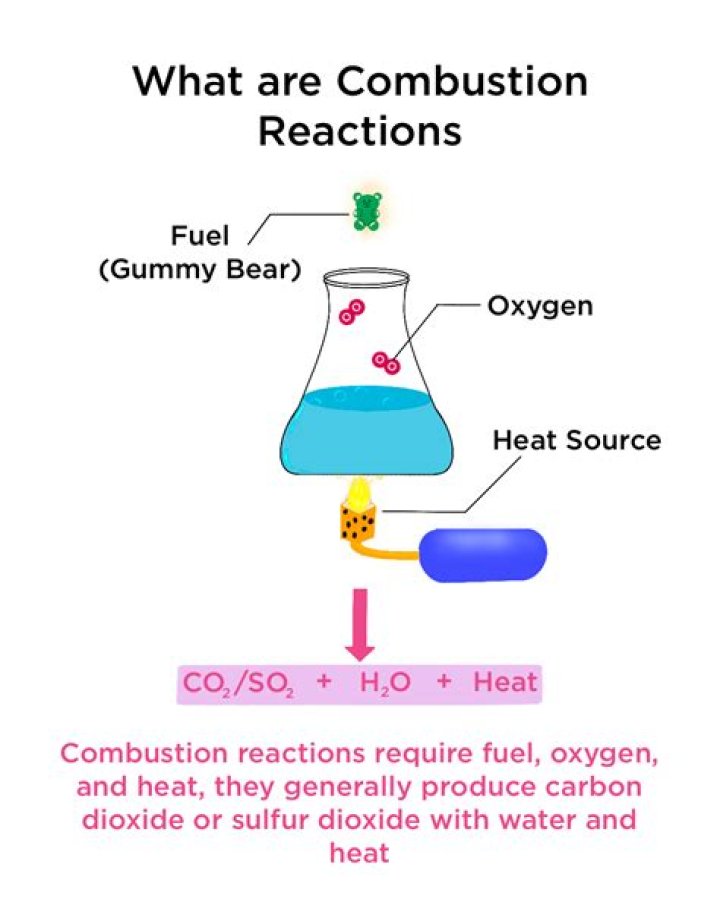
A combustion reaction is when a substance reacts with oxygen and releases a huge amount of energy in the form of light and heat. A combustion reaction always includes a hydrocarbon and oxygen as the reactants and always produces carbon dioxide and water as products.
What are the products of the combustion reaction?
Overview. Combustion reaction is a reaction in which a substance reacts with oxygen gas, releasing energy in the form of light and heat. Combustion reactions must involve O2 as one reactant. The products of the combustion of hydrocarbons are carbon dioxide and water.
How does a combustion reaction occur?
A combustion reaction occurs when a substance reacts quickly with oxygen (O2). For example, in the Figure below, charcoal is combining with oxygen. Combustion is commonly called burning, and the substance that burns is usually referred to as fuel.
Which of the following is an example of combustion of a hydrocarbon?
Hydrocarbon combustion refers to the chemical reaction where a hydrocarbon reacts with oxygen to create carbon dioxide, water, and heat. Hydrocarbons are molecules consisting of both hydrogen and carbon….Carbon dioxide emissions.
| Fuel | kg of CO2 emissions |
|---|---|
| Lignite Coal | 97.9 |
| Subbituminous Coal | 97.4 |
| Diesel | 73.2 |
| Gasoline | 71.5 |
What is the general equation for the combustion of a hydrocarbon?
The reaction typically gives off heat and light as well. The general equation for a complete combustion reaction is: Fuel + O2 → CO2 + H2O.
What are the four products of combustion?
Products of Combustion
- Carbon Dioxide.
- Carbon Monoxide.
- Sulfur Dioxide.
- Nitrogen Oxides.
- Lead.
- Particulate Matter.