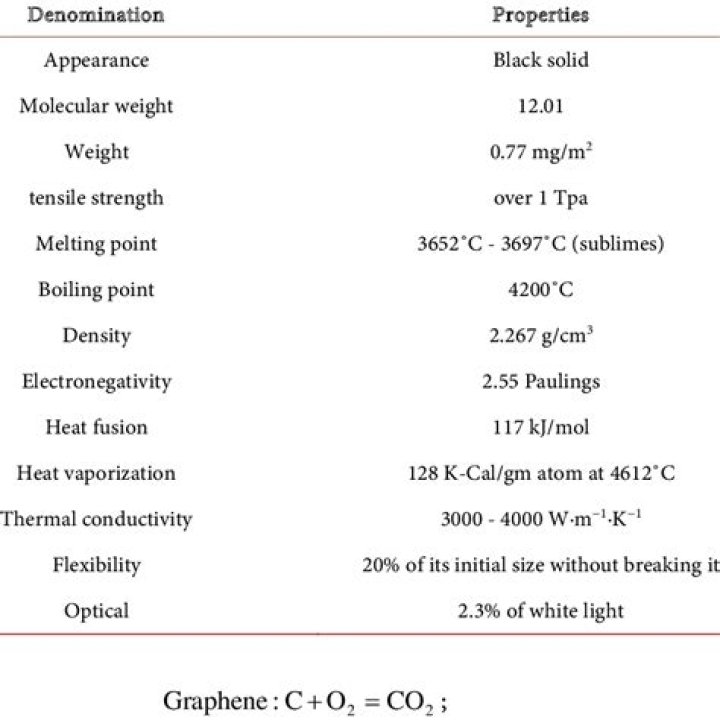
What are 3 physical properties of graphene?
Graphene properties
- High thermal conductivity.
- High electrical conductivity.
- High elasticity and flexibility.
- High hardness.
- High resistance.
- Ionizing radiation is not affected.
- Able to generate electricity by exposure to sunlight.
- Transparent material.
What are the methods of synthesis of graphene?
There are 3 main ways to synthesize graphene, they are: Chemical Vapor Deposition. Chemical or Plasma Exfoliation from natural Graphite. Mechanical cleavage from natural Graphite.
What is synthesis of graphene?
Synthesis of graphene refers to any process for fabricating graphene, depending on the desired size, purity and crystallinity of the final product. Common graphene synthesis approaches include mechanical exfoliation from graphite, chemical vapour deposition, and reduction of graphene oxide through heating.
Can graphene get wet?
Graphene wetting is also important in biological surfaces and designing supercapacitors.” In this study, the researchers investigated the enhanced graphene’s propensity to wet if there is water on the other side of the sheet. They used advanced computer simulations to study this effect at the molecular level.
What is the most useful property of graphene?
electrical conductivity
One of the most useful properties of graphene is that it is a zero-overlap semimetal (with both holes and electrons as charge carriers) with very high electrical conductivity. Carbon atoms have a total of 6 electrons; 2 in the inner shell and 4 in the outer shell.
Why is graphene insoluble?
Graphene won’t dissolve in water or organic solvents. Pure or pristine graphene display poor dispersion due to the strong van der waals force of attraction. Functionalization of graphene (GO or rGO) is the only option to disperse in water or some solvents.
What are the major functions of graphene?
Graphene has a lot of promise for additional applications: anti-corrosion coatings and paints, efficient and precise sensors, faster and efficient electronics, flexible displays, efficient solar panels, faster DNA sequencing, drug delivery, and more.
What are the composition of graphene?
Graphene is a two-dimensional carbon allotrope. It is composed of carbon atoms positioned in a hexagonal design, which can be said to resemble a chicken wire. A single layer of carbon atoms arranged in such a honeycomb structure forms a single graphene sheet.
Why is graphene hydrophobic?
They say that when graphene is exposed to air, a thin layer of hydrocarbon (a compound made entirely of hydrogen and carbon) contaminates its surface and makes it hydrophobic.
Does graphene prevent scratches?
This diamond-like strength enhances the strength of other materials, for instance, graphene has a high abrasion resistance that will protect the paint work from fine marks, swirls and scratches. Its strength also makes it a barrier; since no chemical or material can penetrate its structure.
How strong is graphene compared to diamond?
Strength and stiffness But the atoms within those layers are very tightly bonded so, like carbon nanotubes (and unlike graphite), graphene is super-strong—even stronger than diamond! Graphene is believed to be the strongest material yet discovered, some 200 times stronger than steel.