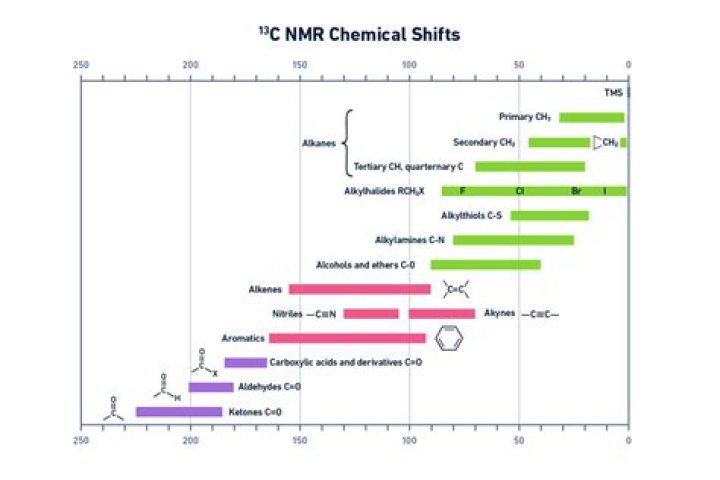
What affects chemical shift in carbon NMR?
C NMR Chemical Shifts The Carbon NMR is used for determining functional groups using characteristic shift values. C chemical shift is affect by electronegative effect and steric effect. The steric effect is observed in acyclic and clyclic system, which leads to downshifted chemical shifts.
What causes Deshielding in C NMR?
There are two major factors that cause different chemical shifts (a) deshielding due to reduced electron density (due electronegative atoms) and (b) anisotropy (due to π bonds). Deshielding: The electrons around the proton create a magnetic field that opposes the applied field.
Where does acetone show up on NMR?
Notes on NMR Solvents
| Solvent | 1H NMR Chemical Shift | 13C NMR Chemical Shift |
|---|---|---|
| Acetone | 2.05 (5) | 206.7 (13) , 29.9 (7) |
| Acetonitrile | 1.94 (5) | 118.7 (1) , 1.39 (7) |
| Benzene | 7.16 (1) | 128.4 (3) |
| Chloroform | 7.26 (1) | 77.2 (3) |
What are common impurities that you might see in your NMR spectrum?
Common impurities that I might see in my NMR spectrum include acetone, water, dichloromethane, methanol, and chloroform. I saw three impurities: methanol, chloroform, and the starting materials. Methanol has a chemical shift of 3.49 ppm.
What is shielding and Deshielding in NMR spectroscopy?
On Professor Hardinger’s website, shielded is defined as “a nucleus whose chemical shift has been decreased due to addition of electron density, magnetic induction, or other effects.” What is Deshielding? Downfield The Nucleus feels stronger magnetic field. Deshielding is the opposite of shielding.
What is shielding and Deshielding effect in NMR?
The peak on the NMR spectrum for this H atom would shift upfield. These H atoms are referred to as being shielded. Chlorine atom is an electronegative atom that will pull the electron density toward it and causes deshielding of the hydrogen nucleus. Therefore, the shift will be to higher ppm.
What is difference between shielding and Deshielding?
Why is ch3 more shielded than ch2?
In ethanol, the electronegative oxygen pulls electron density towards it, with the result that protons near the oxygen get deshielded. So the protons in CH3 are most shielded (upfield), the protons in the CH2 are intermediate, and the H bonded directly to O is least shielded (downfield).
What is TMS peak in NMR?
A peak at a chemical shift of 2.0 is said to be downfield of TMS. The further to the left a peak is, the more downfield it is. Solvents for NMR spectroscopy. NMR spectra are usually measured using solutions of the substance being investigated.