Is sodium an ion?
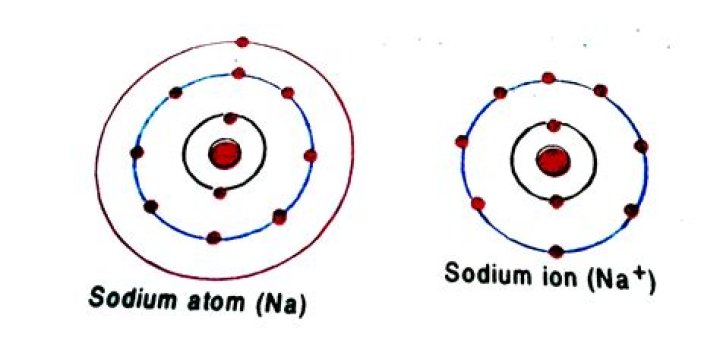
Is sodium an ion?
Sodium(1+) is a monoatomic monocation obtained from sodium. It has a role as a human metabolite and a cofactor. It is an alkali metal cation, an elemental sodium, a monovalent inorganic cation and a monoatomic monocation. Sodium cation is an Osmotic Laxative….4.3Related Element.
| Element Name | Sodium |
|---|---|
| Atomic Number | 11 |
What is more negative K +( potassium or Na +( sodium?
The negative charge within the cell is created by the cell membrane being more permeable to potassium ion movement than sodium ion movement. However, the neurons have far more potassium leakage channels than sodium leakage channels.
Where is sodium ion?
Sodium ions, Na+, and potassium ions, K+, are abundant in the extracellular space and have important roles in the excitability of nerve and muscle cells, and in water and salt homeostasis in biological systems (Hille, 2001; McDonough, 2010; Hodgkin and Katz, 1949).
Is sodium more positive than potassium?
Remember, sodium has a positive charge, so the neuron becomes more positive and becomes depolarized. It takes longer for potassium channels to open. When they do open, potassium rushes out of the cell, reversing the depolarization. Also at about this time, sodium channels start to close.
Is Na+ an anion or cation?
Opposite charges attract each other (positive charge attracts negative charge, and vice versa), so a collection of cations and anions is stable. Neutral sodium atom (Na) becomes sodium cation (Na+) by releasing an electron.
Are sodium ions affected by potassium ions?
The sodium-potassium transporters are individual protein molecules within the pump that transport three sodium ions out of the neuron in exchange for every two potassium ions transported in.
Are chloride ions positive or negative?
The bonds in salt compounds are called ionic because they both have an electrical charge—the chloride ion is negatively charged and the sodium ion is positively charged.
Why is sodium not used in batteries?
Materials. Since the physical and electrochemical properties of sodium differ from those of lithium, the materials generally used for lithium-ion batteries, or even their sodium-containing analogues, are not always suitable for sodium-ion batteries.
Is sodium ion a cation or anion?
By contrast, atoms of calcium, magnesium, aluminum, and sodium tend to lose electrons and form cations. Sodium chloride—what we know as table salt—is actually composed of an anion and a cation (written as NA+CL-).
Why is sodium a positive ion?
An ion is a charged particle, such as Na+, the sodium ion. It has a positive charge, because it is missing one electron. Other ions, of course, are negatively charged.
What are sodium ions used for in the body?
Sodium is both an electrolyte and mineral. It helps keep the water (the amount of fluid inside and outside the body’s cells) and electrolyte balance of the body. Sodium is also important in how nerves and muscles work.



