Is HCN polar or non polar?
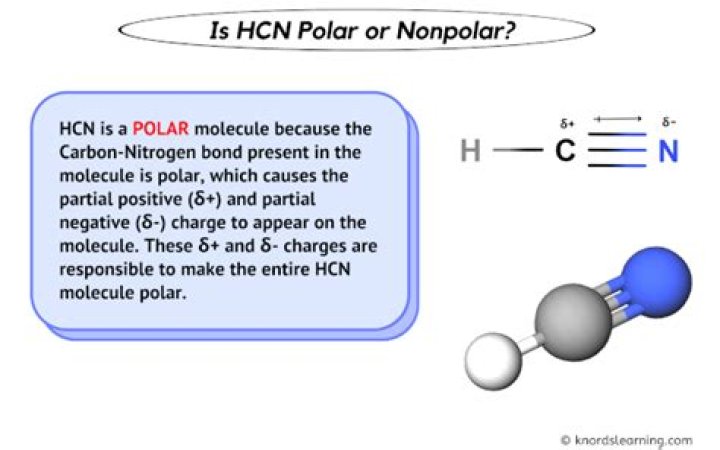
Is HCN polar or non polar?
HCN, or hydrogen cyanide, is a polar molecule because there is a large electronegative difference between the N and H across the linear molecule. It consists of two polar bonds whose polarities line up in the same direction. The Lewis structure determines whether an entire molecule is polar or essentially nonpolar.
Why is the molecule polar Quizizz?
Why is the molecule polar? There is a nonbonding pair on the central atom. There are different types of elements bonded to the central atom. There are no nonbonding pairs on the central atom and all of the atoms bonded to the central atom are the same.
What is the bond type of HCN?
triple covalent bond
In HCN, Carbon is bonded to Nitrogen with a triple covalent bond consisting of one sigma bond and two pi bonds. The sigma bond is formed by overlapping hybridized orbitals, with the two remaining unhybridized orbitals overlapping to form the two pi bonds.
What is the electronegativity of HCN?
HCN is a linear molecule with a single bond between the H and the C and a triple bond between the C and the N. Looking at the electronegativities H(2.1<2.5<3.0). Carbon will pull the shared electrons in the H-C bond toward C and nitrogen will pull the shared electrons in the C-N triple bond toward N.
What is the electron geometry of HCN?
Linear
HCN
| Central atom: | C |
|---|---|
| Total VSEP: | 4 |
| 1 x triple bond: | − 2 pairs |
| Revised Total: | 2 |
| Geometry: | Linear |
Is trigonal planar polar or NonPolar?
Trigonal Planar Molecular Geometry
| Shape: | trigonal planar |
|---|---|
| Lone Pairs: | 0 |
| Polar/NonPolar: | NonPolar |
| Hybridization: | sp2 |
| Examples: | BF3, CO32- |
Is HCN bent or linear?
Hydrogen cyanide is a linear molecule. A Lewis formulation counts 1 electron from the hydrogen, 4 electron from the carbon, and 5 electron from the nitrogen, so 5 electron pairs to distribute.
How do you find the electronegativity difference in HCN?
For HCN you will calculate the difference between H and C which is 0.4 and the difference between C and N which is 0.5. To determine if a molecule overall is polar or nonpolar you must look at both the electronegativity differences in each bond and the shape of the molecule.
Is cf2h2 polar or nonpolar?
CH2F2 is polar. The central atom is Carbon. It has 4 atoms bonded to it; thus, it has a steric number of 4.