Is breaking bond endothermic?
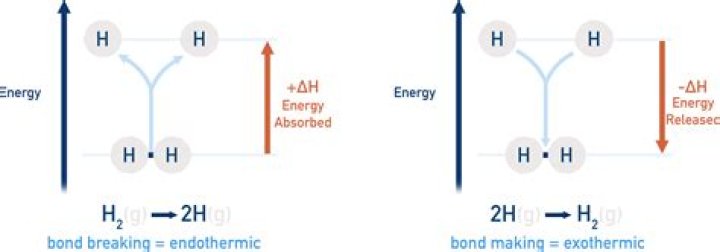
Bond breaking and bond forming occurs during a chemical reaction. The energy changes in chemical reactions are caused by bond breaking and bond forming. Breaking a bond is endothermic. Energy is taken in to break a chemical bond.
Why does breaking bonds release energy?
The reason there is energy released in the process is because the products formed (ADP and hydrogenphosphate/phosphate) have stronger covalent bonds (plus intermolecular forces with the surrounding solution and dissolved ions) than the starting materials. This is the case for any exothermic process.
Is breaking bonds positive or negative?
Generally, a positive change in enthalpy is required to break a bond, while a negative change in enthalpy is accompanied by the formation of a bond. In other words, breaking a bond is an endothermic process, while the formation of bonds is exothermic.
Is breaking bonds Endergonic or Exergonic?
In other words, there is a net release of free energy. Breaking chemical bonds in the reactants releases more energy than that used to form new chemical bonds in the products. Exergonic reactions are also known as exoergic, favorable, or spontaneous reactions.
Is breaking the bond of i2 exothermic?
The bond breaking is always endothermic. The formation of new bonds is exothermic, so depending on whether the old bonds or the new bonds were stronger, the reaction overall can be either endothermic or exothermic.
Why is a reaction exothermic in terms of bonds?
Chemical reactions that release energy are called exothermic. In exothermic reactions, more energy is released when the bonds are formed in the products than is used to break the bonds in the reactants. Exothermic reactions are accompanied by an increase in temperature of the reaction mixture.
What happens to energy when bonds are broken?
Breaking chemical bonds absorbs energy, while making new bonds releases energy, with the overall chemical reaction being endothermic or exothermic.
Does the breaking of bonds in ATP release energy?
hydrolysis
When one phosphate group is removed by breaking a phosphoanhydride bond in a process called hydrolysis, energy is released, and ATP is converted to adenosine diphosphate (ADP). Likewise, energy is also released when a phosphate is removed from ADP to form adenosine monophosphate (AMP).
When a bond is broken the bond energy is positive because heat?
It is important to note that the breaking of a chemical bond is always an endothermic process (because energy must be supplied to the molecule in order to break the chemical bonds that constitute it). Thus, the enthalpy change associated with the breaking of a chemical bond is always positive (ΔH > 0).
Is breaking bonds Endergonic?
Forming a bond is always an exothermic reaction because it releases energy. Breaking a bond always requires energy, and is thus an endothermic process.
Do bonds form in endergonic reactions?
Endergonic reactions absorb energy from their surroundings. The chemical bonds that are formed from the reaction are weaker than the chemical bonds that were broken. The free energy of the system increases. The change in the standard Gibbs Free Energy (G) of an endergonic reaction is positive (greater than 0).
Is breaking a hydrogen bond exothermic?
Bond formation is an EXOTHERMIC process. Bond breaking (i.e. fission, cleavage) is an ENDOTHERMIC process.