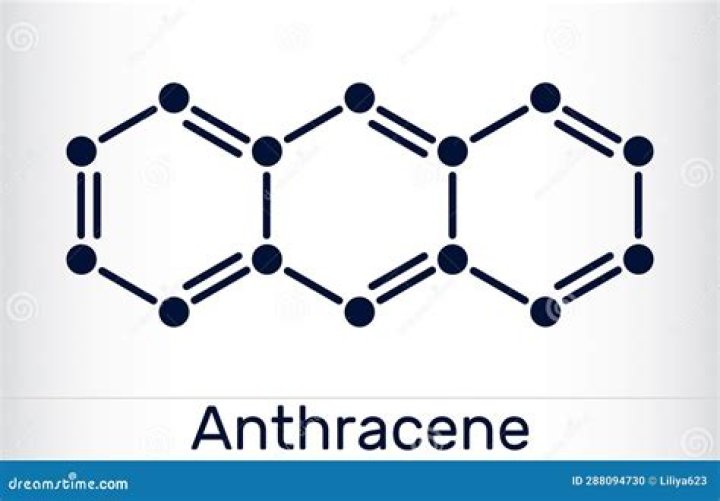
Is anthracene a carcinogen?
The US Environmental Protection Agency weight-of-evidence classification for benz[a]anthracene is B2, a probable human carcinogen, for both oral and inhalation exposure based on adequate animal evidence and no human evidence.
Is anthracene an irritant?
* Breathing Anthracene can irritate the nose, throat and lungs causing coughing and wheezing. * Eye contact can cause irritation and burns. * Anthracene may cause a skin allergy. If allergy develops, very low future exposure can cause itching and a skin rash.
What causes fluorescence in anthracene?
Anthracene is colorless but exhibits a blue (λmax = 400–450 nm) fluorescence under ultraviolet (UV) light. In contaminated aquatic environments, toxic response of anthracene is aggravated by sunlight. Anthracene undergoes photodimerization by UV light.
Which of the following methods is used for the synthesis of anthracene?
A classic laboratory method for the preparation of anthracene is by cyclodehydration of o-methyl- or o-methylene-substituted diarylketones in the so-called Elbs reaction.
What does anthracene do to humans?
In humans, anthracene may cause acute dermatitis with symptoms of burning, itching, and edema. Prolonged dermal exposure produces pigmentation, cornification of skin surface layers, and telangiectasis. Anthracene is photosensitizing, potentiating skin damage elicited by exposure to ultraviolet radiation.
What is anthracene used for?
anthracene, a tricyclic aromatic hydrocarbon found in coal tar and used as a starting material for the manufacture of dyestuffs and in scintillation counters. Crude anthracene crystallizes from a high-boiling coal-tar fraction. It is purified by recrystallization and sublimation.
Is anthracene poisonous?
Anthracene, also referred to as paranaphthalene or green oil, is a polycyclic aromatic hydrocarbon (PAH). Although a large body of literature exists on the toxicity and carcinogenicity of other PAHs, toxicity data for anthracene are limited. Information on the toxicity of anthracene exposure in humans is very limited.
Is anthracene a VOC?
Semivolatile organic compounds (SVOCs) are a subgroup of VOCs that tend to have a higher molecular weight and higher boiling point temperature….The following analytes are commonly reported:
| N-nitrosodimethylamine | 2,4-dinitrotoluene |
|---|---|
| hexachlorobutadiene | benzo(a)anthracene* |
| 4-chloro-3-methylphenol | chrysene* |
Where is anthracene used?
It is primarily used as an intermediate in the production of dyes, smoke screens, scintillation counter crystals, and in organic semiconductor research. Although a large body of literature exists on the toxicity of PAHs, data for anthracene are limited.
Does anthracene undergo addition reaction?
The correct answer is option D.