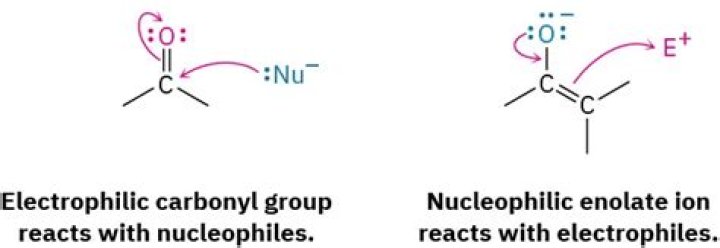
Is a carbonyl an electrophile?
Electrophile is another term for Lewis acid. Lewis acids attract electrons. Lewis acids have a positive charge on an atom, a partial positive charge on an atom, or an atom lacking an octet. Carbonyl compounds are good electrophiles.
How is a carbonyl group an electrophile?
Because oxygen is more electronegative than carbon, carbonyl compounds often have resonance structures which affect their reactivity. This relative electronegativity draws electron density away from carbon, increasing the bond’s polarity, therefore making carbon an electrophile (i.e. slightly positive).
Is a carbonyl an electrophile or nucleophile?
The carbonyl carbon is an electrophile, and it reacts with nucleophiles to produce a tetrahedral species. It’s the fundamental reaction of carbonyl chemistry. Now, if you want to make a carbonyl even HUNGRIER for nucleophiles, you can add an acid to it.
Which carbonyl is most electrophilic?
The stabilization afforded by the carbonyl group depends on how electron-poor it is. Placing a highly electronegative grouping like CF3 adjacent to the carbonyl makes the carbonyl much more electrophilic, which makes it better able to stabilize negative charge.
Which class of compound is an example of carbonyl compound?
Examples of class 1 carbonyl compounds are amides, esters, carboxyllic acids and anhydrides. Class 2 compounds have carbonyl compounds that do not leave the molecule when reacting, examples of this type of carbonyl compound are ketones and aldehydes.
Is amide a carbonyl compound?
In amides, the carbonyl carbon is bonded to a nitrogen. The nitrogen in an amide can be bonded either to hydrogens, to carbons, or to both. Another way of thinking of an amide is that it is a carbonyl bonded to an amine. In esters, the carbonyl carbon is bonded to an oxygen which is itself bonded to another carbon.
Which class of compounds is an example of a carbonyl compound?
The group is a constituent of carboxylic acids, esters, anhydrides, acyl halides, amides, and quinones, and it is the characteristic functional group (reactive group) of aldehydes and ketones. Carboxylic acids (and their derivatives), aldehydes, ketones, and quinones are also known collectively as carbonyl compounds.
Which class of compound is an example of a carbonyl compound Mcq?
The various classes of organic compounds containing carbonyl groups are aldehydes, ketones, carboxylic acids and their derivatives. The carbonyl carbon of the simplest aldehyde, formaldehyde is bonded to two hydrogen atoms.
What atoms are Electrophiles?
Because electrophiles accept electrons, they are Lewis acids. Most electrophiles are positively charged, have an atom that carries a partial positive charge, or have an atom that does not have an octet of electrons….Electrophilicity scale.
| Fluorine | 3.86 |
|---|---|
| Sulfur dioxide | 2.01 |
| Carbon disulfide | 1.64 |
| Benzene | 1.45 |
| Sodium | 0.88 |
Are aldehydes Electrophiles?
Aldehydes are electrophilic compounds to which humans are pervasively exposed. In contrast, α,β-unsaturated carbonyl derivatives, alkenals, and the α-oxoaldehydes are soft electrophiles that preferentially react with soft nucleophilic thiolate groups on cysteine residues.
Which organic compounds are described as Carbonyls?
Carboxylic acids (and their derivatives), aldehydes, ketones, and quinones are also known collectively as carbonyl compounds.
What are carbonyl compounds give Example Class 12?
The carbonyl group in aldehydes is at the end of the carbon chain, while the carbonyl group in ketones is in the centre of the carbon ring. Propanol, butanol, 4-chlorobutanol, and other aldehydes are examples of aldehydes, while propanone, acetone, 2-methyl-3-pentanone, and other ketones are examples of ketones.