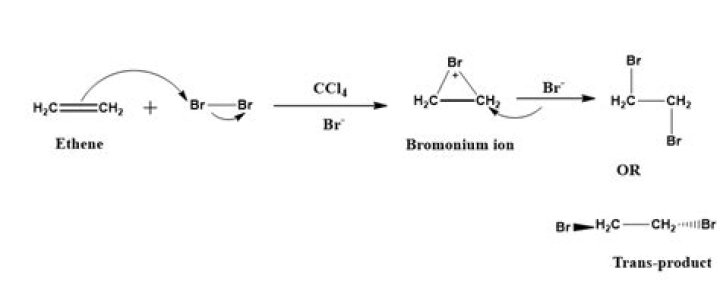
How would you test for the presence of ethene?
If you have a gaseous alkene like ethene, you can bubble it through either pure liquid bromine or a solution of bromine in an organic solvent like tetrachloromethane. The reddish-brown bromine is decolourised as it reacts with the alkene.
How do you test for an alkene?
A simple test with bromine water can be used to tell the difference between an alkane and an alkene. An alkene will turn brown bromine water colourless as the bromine reacts with the carbon-carbon double bond.
How do you test ethane and ethene?
You can distinguish ethane from ethene by adding a drop of bromine water to both samples. One will turn from brown to colorless (ethene). The bromine adds to the C-C double bond in ethene in an addition reaction and gets used up so the color goes away.
How can you distinguish between ethane and ethene using a chemical test?
Take ethane and ethene in two separate test tubes and dissolve them in carbon tetrachloride solution. Pass bromine gas into the two test tubes. If the color of bromine gas is discharged, then that gas is ethene and if the color of gas remains the same, then that test tube contains ethane gas.
Which compound give bromine water test positive?
Enols, alkenes, aniline, glucose, phenols and acetyl groups are the most common compounds to undergo bromine water test. The test also identifies the presence of an aldehyde group in the compound.
How will you distinguish between ethene and ethyne?
Ethene is the IUPAC name for Ethylene. Ethyne is the IUPAC name for Acetylene. The main difference between Ethene and Ethyne is that Ethene is composed of sp2 hybridized carbon atoms whereas Ethyne is composed of sp hybridized carbon atoms.
How can you distinguish between ethane gas and Ethylene gas?
Ethane has single bonds only, but ethene has a double bond. Therefore, ethane is considered as a saturated hydrocarbon, whereas ethene is considered as an unsaturated hydrocarbon. When naming the alkenes like ethene, “ene” is used instead of “ane” in the end of the alkane name (ethane).
How do you distinguish between ethanol and ethene?
It’s a significant one, as both are different types of compound. Ethanol is an aliphatic alcohol (CH3-CH2-OH) with a hydroxyl group attached to one of the central carbon atoms; it is liquid at room temperature & pressure. Ethene by contrast is an unsaturated hydrocarbon with a carbon-carbon double bond and is a gas.
What type of reaction is bromine water test?
Bromine water test (saturation test) The bromine water test is a qualitative test, used to identify the alkene or alkane functional groups present in the compound. Alkene groups react with bromine water in the dark condition and undergo an addition reaction, to give a decolourized solution.
What’s the best way to test for ethene?
How do you test for ethene? In laboratory you can test it with bromine water (red-brown) which decolorizes when you’ll bubble out the gas from it. At home you can test it with some fruits and vegitables because it’s a ripening agent. You may place unripe tomatoes, bananas or mangoes in ethene gas they will start ripening quickly.
How to test if ethene gas is produced in an experiment?
In laboratory you can test it with bromine water (red-brown) which decolorizes when you’ll bubble out the gas from it. At home you can test it with some fruits and vegitables because it’s a ripening agent. You may place unripe tomatoes, bananas or mangoes in ethene gas they will start ripening quickly.
What foods can you test for ethene gas?
At home you can test it with some fruits and vegitables because it’s a ripening agent. You may place unripe tomatoes, bananas or mangoes in ethene gas they will start ripening quickly. Click to see full answer. Also asked, how do you test for ethane?
How can we identify ethane from ethene?
So they can be identified by their displayed formulae, as ethene has the double bond functional group. Chemically, they can be distinguished most easily by the reaction with bromine water, which is orange/red in colour. Add a few drops to ethene and bromine water is decolourised as 1,2-dibromoethane, a saturated compound forms.
How do you test for ethene? In laboratory you can test it with bromine water (red-brown) which decolorizes when you’ll bubble out the gas from it. At home you can test it with some fruits and vegitables because it’s a ripening agent. You may place unripe tomatoes, bananas or mangoes in ethene gas they will start ripening quickly.
In laboratory you can test it with bromine water (red-brown) which decolorizes when you’ll bubble out the gas from it. At home you can test it with some fruits and vegitables because it’s a ripening agent. You may place unripe tomatoes, bananas or mangoes in ethene gas they will start ripening quickly.
At home you can test it with some fruits and vegitables because it’s a ripening agent. You may place unripe tomatoes, bananas or mangoes in ethene gas they will start ripening quickly. Click to see full answer. Also asked, how do you test for ethane?
So they can be identified by their displayed formulae, as ethene has the double bond functional group. Chemically, they can be distinguished most easily by the reaction with bromine water, which is orange/red in colour. Add a few drops to ethene and bromine water is decolourised as 1,2-dibromoethane, a saturated compound forms.