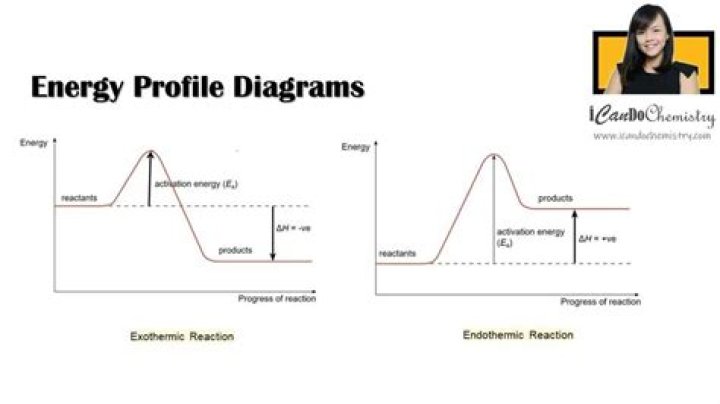
How the potential energy diagram is used to identify if the reaction is endothermic or exothermic?
The energy changes that occur during a chemical reaction can be shown in a diagram called a potential energy diagram, or sometimes called a reaction progress curve. Recall that the enthalpy change (ΔH) is positive for an endothermic reaction and negative for an exothermic reaction. …
Where is enthalpy on a potential energy diagram?
In the context of a potential energy diagram, there is an enthalpy of bond formation at the equilibrium bond distance. It is not entirely obvious, but it is the potential energy shown on the y-axis.
Is combustion of octane exothermic?
The combustion of methane or octane is exothermic; it releases energy.
Is enthalpy of combustion exothermic?
Combustion is an oxidation reaction that produces heat, and it is therefore always exothermic. All chemical reactions first break bonds and then make new ones to form new materials. Breaking bonds takes energy while making new bonds releases energy.
What is the combustion reaction of octane?
C8H18 + 25/2 O2 —> 8CO2 + 9 H2O. To obtain a equation containing whole numbers, we multiply the entire equation by 2. This gives the final equation. 2 C8H18 + 25 O2 —> 16 CO2 +18 H2O.
Is the combustion reaction of butane exothermic or endothermic?
Combustion Reaction Butane releases its chemical energy by undergoing hydrocarbon combustion. Below is a hydrocarbon combustion animation showing the net reaction that occurs when butane combines with oxygen. The hydrocarbon combustion reaction releases heat energy and is an example of an exothermic reaction.
How is combustion an exothermic reaction?
Overall, combustion is an exothermic reaction given off or exiting , which means that energy is released. Usually, heat and light are released during a combustion reaction. During exothermic reactions (like combustion), bonds are broken, which allows the energy trapped in the bonds to be released and do work.
Is heat of combustion and enthalpy of combustion same?
Standard enthalpy of combustion (ΔH∘C Δ H C ∘ ) is the enthalpy change when 1 mole of a substance burns (combines vigorously with oxygen) under standard state conditions; it is sometimes called “heat of combustion.” For example, the enthalpy of combustion of ethanol, −1366.8 kJ/mol, is the amount of heat produced when …