How many valence electrons does carbon bonding have?
When bonded with a full octet (such as in methane) carbon has eight valence electrons (two per covalent bond).
Are bonding electrons the same as valence electrons?
Valence electrons are outer shell electrons with an atom and can participate in the formation of chemical bonds. In single covalent bonds, typically both atoms in the bond contribute one valence electron in order to form a shared pair.
How many electrons does carbon bond?
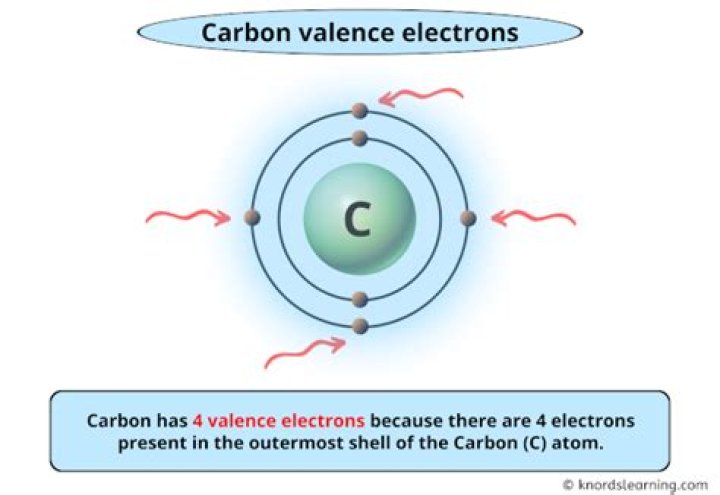
four electrons
Carbon contains four electrons in its outer shell. Therefore, it can form four covalent bonds with other atoms or molecules.
Why can carbon only form 4 bonds?
Carbon has 6 electrons, two in its inner shell and four in its valence shell. When carbon takes four electrons from other atoms, in which it forms ionic bonds, it has a full valence shell, so it is unable to from any more bonds.
Does carbon have 5 valence electrons?
Like other group 14 elements, carbon has four valence electrons. Valence electrons are the electrons in the outer energy level of an atom that are involved in chemical bonds.
How many covalent bonds can carbon form?
four covalent bonds
One carbon atom forms four covalent bonds with four hydrogen atoms by sharing a pair of electrons between itself and each hydrogen (H) atom.
What is the relationship between valence electrons and covalent bonding?
Valence electrons are the electrons in the outermost shell of the atom and covalent bonding is the sharing of the atom’s valence electrons with another atom.
Does carbon have 2 or 4 electrons?
So… for the element of CARBON, you already know that the atomic number tells you the number of electrons. That means there are 6 electrons in a carbon atom. Looking at the picture, you can see there are two electrons in shell one and four electrons in shell two. ► More about the history and places to find carbon.
How many valence electrons does carbon share in order to complete its valence shell?
4 electrons
Carbon (4 electrons in the valence shell) combines with four hydrogen atoms to form a stable covalent compound where it shares 8 electrons, while each hydrogen shares 2. Thus every atom in this stable molecule fulfills the octet rule.
Which bonding is not possible for carbon with its 4 valence electrons?
There is no 4 bond formed between carbon because of the carbon electron orbitals. Since it has 4 valence electrons, it needs 4 more to electrons to fill its outer energy level. It does so by forming covalent bonds with another element, in order to complete its Octet rule.
What type of bonds are formed by carbon?
Carbon forms covalent bonds with atoms of carbon or other elements. There is a great diversity of carbon compounds, ranging in size from just one to thousands of atoms. Carbon has four valence electrons, so it can achieve a full outer energy level by forming four covalent bonds.