How many stereoisomers are possible for Ribitol?
four possible stereoisomers
There are four possible stereoisomers.
Is Ribitol a meso compound?
Ribitol (1, Fischer projection) is a meso polyol that is optically inactive. A plane of symmetry passes through C3 and its hydrogen and hydroxyl group.
How is Ribitol formed?
Ribitol, or adonitol, is a crystalline pentose alcohol (C5H12O5) formed by the reduction of ribose. It occurs naturally in the plant Adonis vernalis as well as in the cell walls of some Gram-positive bacteria, in the form of ribitol phosphate, in teichoic acids….CHEBI:15963 – ribitol.
| ChEBI Name | ribitol |
|---|---|
| Download | Molfile XML SDF |
What are the 2 isomers of glucose?
There are two enantiomers of glucose, called D-glucose and L-glucose. The D-enantiomer is the common sugar that our bodies use for energy.
What is Chirotopic?
What is Chirotopic? The description of an atom (or point, group, face, etc. in a molecular model) that resides within a chiral environment. One that resides within an achiral environment has been called achirotopic.
Is xylitol optically active?
It is even possible to have diastereomeric pairs in which neither member is optically active. and xylitol. They are diastereomers of each other, but they each have an internal plane of symmetry. They are both meso compounds, and they are both optically inactive.
Is Ribitol soluble in water?
It contributes to the chemical structure of riboflavin and flavin mononucleotide. Solubility : Soluble in water (100 mg/ml).
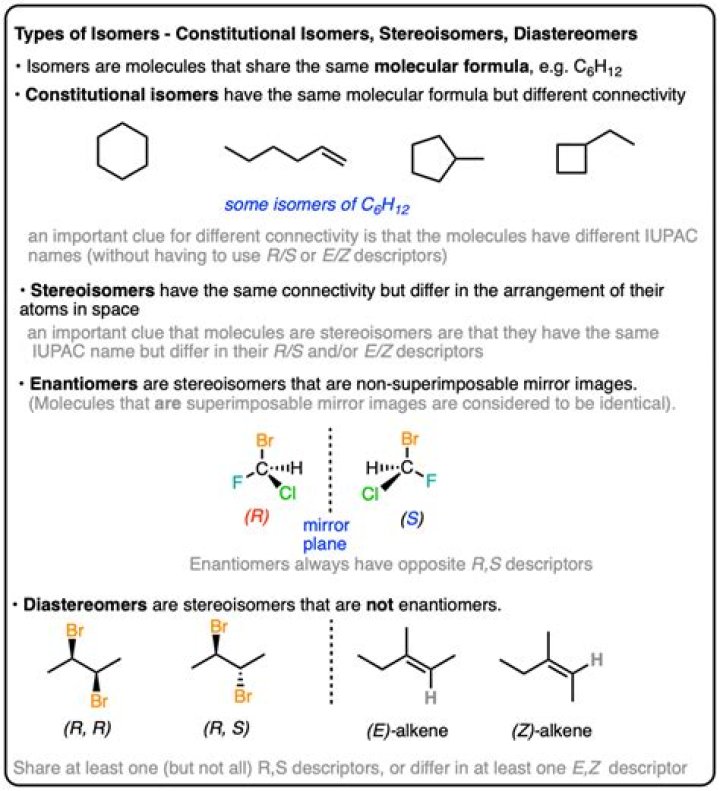
Are stereoisomers structural isomers?
Structural isomers have the same molecular formula but a different bonding arrangement among the atoms. Stereoisomers have identical molecular formulas and arrangements of atoms. They differ from each other only in the spatial orientation of groups in the molecule.
What is a stereoisomer of glucose?
There are two enantiomers of glucose, called D-glucose and L-glucose. The D-enantiomer is the common sugar that our bodies use for energy. It has n = 4 stereocenters, so therefore there are 2 n = 2 4 = 16 possible stereoisomers (including D-glucose itself).
What is a stereoisomer example?
Stereoisomers have the same molecular formula, and the same connectivity except for the arrangement in 2D or 3D space. For example, cis- and trans-but-2-ene both contain 2 CH3- groups, 2 H- and a C=C. Both can be represented generically as CH3CH=CHCH3 (i.e. same connectivity).