How many moles are in P4O10?
Explanation: 1 mol P4O10 contains 4 mol P .
How many moles of phosphorus atoms are there in 4 moles of P4O10?
There is four moles off phosphorus, right? Say 4 to 1 mole ratio. And once you multiply the moles of the compound by the moles off the phosphorus, you will get an answer of 114 moles off phosphorus. Okay, so in one move off of that compound, you have four moles of phosphorus.
How many moles of n are in .197 g n2o?
There are 0.00896 moles of N.
What is the molar mass of P4O10?
141.944 g/mol
Phosphorus pentoxide/Molar mass
How many moles of oxygen are in P4O10?
mole) = 0.2641 moles [the g cancles and moles moves to the top]. Rounded to 2 significant figures (since 75 is only 2 significant figures), the number of moles of P4O10 is 0.26. The equations says we need 5 moles of O2 for every mole of P4O10.
How many phosphorus atoms does P4O10?
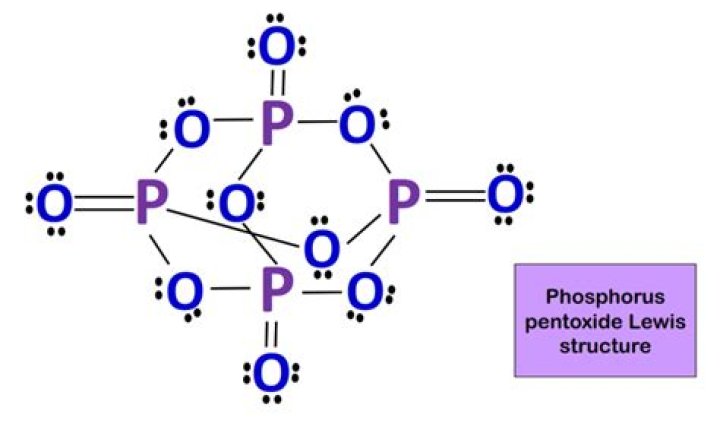
Phosphorous pentoxide is the common name for the compound with a chemical formula P4O10. Phosphorus pentoxide is a covalent compound that is composed of 4 phosphorus (P) atoms and 10 oxygen (O) atoms.
How many P atoms are present in P4O10?
4 phosphorus atoms
In ${P_4}{O_{10}}$, there are 4 phosphorus atoms and 10 oxygen atoms.
How many moles of n are in 0.167 g of n2o0 167 g N2O?
Moles of N2O = 0.167⋅g44.01⋅g⋅mol−1=3.80×10−3⋅mol . And the molar quantity of nitrogen ATOMS, is twice this molar quantity: Moles of nitrogen atoms=2×3.80×10−3⋅mol=7.60×10−3⋅mol………
How many moles of NN are in 0.219 g of n2o0 219 g of N2O?
1 Approved Answer Molar mass N2O = 44.0129 g/mol 0.219 g = 0.219 /44.0129 = 0.0049mol N2O You ask for mol N which I take to mean moles of nitrogen…
How many total atoms are in the formula P4O10?
How many total atoms are in the formula P4O10? For every molecule of P4O10, there are 14 atoms (four phosphorus, ten oxygen).
What is the molar mass of ph3?
33.99758 g/mol
Phosphine/Molar mass
How many particles are in 7.5 moles?
Explanation: And Avogadro’s number , 6.022×1023⋅mol−1 specifies a molar quantity…… And so 7.5⋅mol×6.022×1023⋅mol−1≅4.5×1024 sulfur atoms .