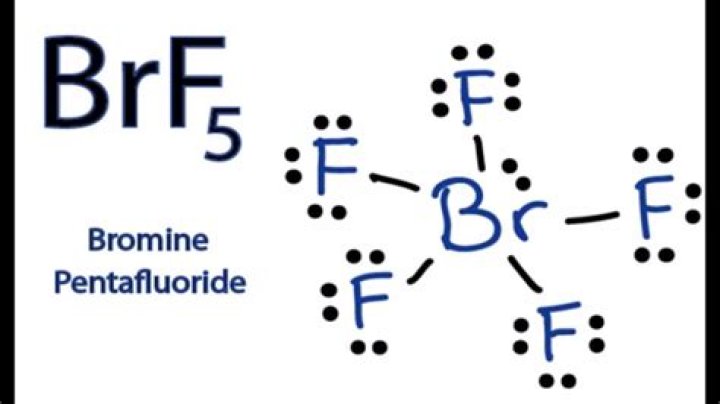
How many electron pairs are in BrF5?
five
In the structure, bromine has one lone pair and five bonds. Each covalent bond contains a pair of electrons, which means the total number of electrons around the central atom is 12 electrons. Hence, there are 6 electron groups present around the central atom in bromine pentafluoride.
What type of bond is BrF5?
Hybridization of BrF5 (Bromine Pentafluoride)
| Name of the Molecule | Bromine Pentafluoride |
|---|---|
| Molecular Formula | BrF5 |
| Hybridization Type | sp3d2 |
| Bond Angle | 90o |
| Geometry | Square Pyramidal |
What is the dipole moment of BrF5?
Does BrF5 have a dipole moment? (BrF5), or bromine pentafluoride has a square pyramidal structure as in the first figure. If we consider PCl5 which has a trigonal bi-pyramidal structure, there is no net dipole moment as the molecule is completely symmetric around the central phosphorus atom.
What is the electron geometry of BrF5 enter the electron geometry of the molecule?
Thus, the electron geometry of BrF5 is octahedral.
Is BrF5 follow octet rule?
For the BrF5 Lewis structure the total number of valence electrons (found on the periodic table) is 42. Note that in the Lewis structure for BrF5, Bromine (B) is in Period Four on the periodic table. This means it can hold more than eight valence electrons. This is called an expanded octet.
Does BrF5 follow octet rule?
Is BrF5 dipole dipole?
Starts here2:06Is BrF5 Polar or Nonpolar? (Bromine pentafluroide) – YouTubeYouTube
What is the electron geometry of BrF5 chegg?
The electron pair geometry is octahedral.
What is electron domain geometry of BrF5?
The hybridization of BrF5 is Sp³d². Bromine pentafluoride is polar in nature. The molecular geometry of BrF5 is square pyramidal and its electron geometry is octahedral. The bond angle of BrF5 is 90º.
How many bonds in BrF5 have dipole?
Because of its asymmetric shape of the BrF5 molecule, BrF5 is a polar molecule because it creates unequal charge distribution surrounding atoms, making it difficult to cancel out the dipole along with five Br-F bonds with them, resulting in a net dipole moment for it.