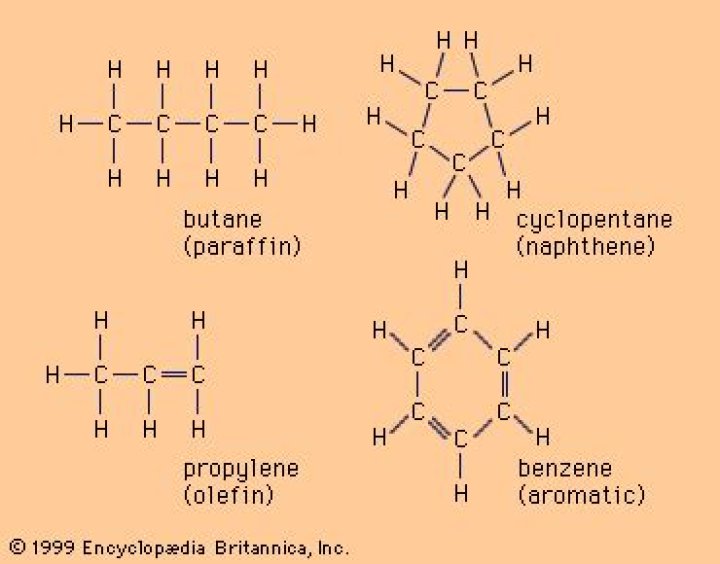
How long can a hydrocarbon chain be?
The inner tube has a diameter of about 0.7 nm. An international research team has created the longest linear carbon chain to date by linking more than 6,000 carbon atoms into a single-file strand that stretches for nearly a micrometer (Nat.
What is the length of carbon chain?
The bond lengths of carbon chains characterizes the type of bond between the carbons, for example alkanes have a bond length of 1.53 Å , alkenes 1.33 Å and alkynes 1.20 Å [10] . In chemistry, two useful types of carbon chain structures are cumulenes and polyynes.
How does the chain length of a hydrocarbon affect its properties?
As the hydrocarbon chain length increases, viscosity increases. As the hydrocarbon chain length increases, flammability decreases. hydrogen in the fuels are oxidised, releasing carbon dioxide, water and energy. The boiling point of the chain depends on its length.
How can long chain hydrocarbons turn into short hydrocarbons?
Cracking allows large hydrocarbon molecules to be broken down into smaller, more useful hydrocarbon molecules. Fractions containing large hydrocarbon molecules are heated to vaporise them. Watch this practical demonstration of cracking alkanes in the lab. Cracking produces a mixture of smaller alkanes and alkenes.
What is hydrocarbon chain?
A hydrocarbon chain is an organic molecule consisting of nothing else but carbon and hydrogen atoms arranged in a chain. And each carbon atom in the chain is bonded to one or up to three hydrogen atoms. Hydrocarbon chains are non-polar and therefore would not readily mix with polar molecules such as water.
Why are shorter chain hydrocarbons more flammable?
Shorter hydrocarbon molecules have weaker intermolecular forces and lower boiling points. They are highly volatile and therefore extremely flammable.
What is the difference between long chain and short chain hydrocarbons?
Long chain hydrocarbons are split into a mixture of smaller alkanes and alkenes. The length of the hydrocarbon chain determines its properties. A shorter hydrocarbon is less viscous, more flammable, more volatile and has a lower boiling point than a long chain hydrocarbon.