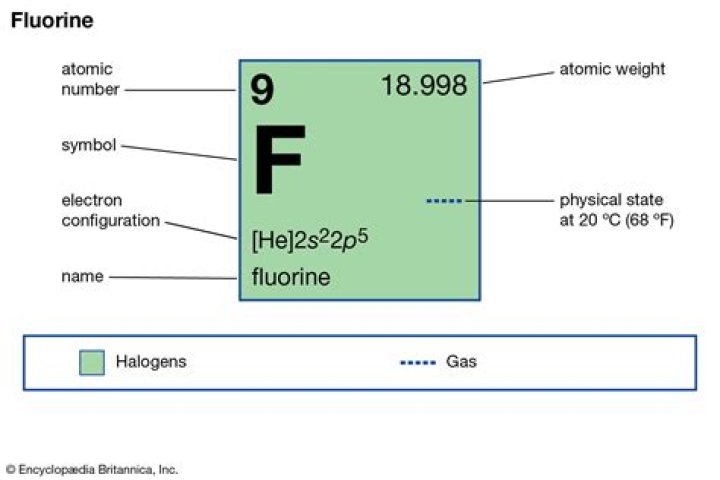
How is pure fluorine made?
The most common fluorine minerals are fluorite, fluorspar and cryolite, but it is also rather widely distributed in other minerals. It is the 13th most common element in the Earth’s crust. Fluorine is made by the electrolysis of a solution of potassium hydrogendifluoride (KHF2) in anhydrous hydrofluoric acid.
What is fluorine pure form?
At room temperature, fluorine is a gas of diatomic molecules, pale yellow when pure (sometimes described as yellow-green). Fluorine condenses into a bright yellow liquid at −188 °C (−306 °F), a transition temperature similar to those of oxygen and nitrogen. Fluorine has two solid forms, α- and β-fluorine.
Can fluorine be found pure?
1886
Fluorine/Discovered
How is fluorine mined?
The main mining areas for fluorite are China, Mexico and Western Europe. Fluorine occurs naturally in the earth’s crust where it can be found in rocks, coal and clay. Fluorides are released into the air in wind-blown soil. Fluorine is the 13th most aboundant element in the Earth’s crust: 950 ppm are contanined in it.
How dangerous is fluorine?
Fluorine is an extremely strong irritant to all tissues it comes in contact with. It can cause injury ranging from mild irritation to caustic burns depending on the concen- tration of the gas at the time of exposure. It is a very severe irritant of the lungs, mucous membranes, skin and eyes.
What are 5 uses of fluorine?
Uses of Fluorine
- Molecular fluorine and Atomic fluorine are used in semiconductor manufacturing for plasma etching, MEMs fabrication, and flat panel display production.
- Chlorofluorocarbons are used extensively used in air conditioners and refrigerators.
- Fluorides are also added to toothpaste to prevent dental cavities.
Can fluorine kill you?
In its gaseous form, fluorine is incredibly dangerous. Small amounts of fluorine gas can case eye and nose irritation while larger amounts can be fatal, according to Lenntech. Hydrofluoric acid, as another example, can also prove to be fatal when even a small splash on the skin occurs, according to Chemicool.
What are 3 uses of fluorine?
What are 3 uses for fluorine?
Uses of Fluorine Compounds of fluorine, including sodium fluoride, are used in toothpaste and in drinking water to prevent dental cavities. Hydrofluoric acid can dissolve glass and is used to etch the glass in light bulbs and in other products.
Do bananas have fluoride?
Most raw fruit has fluoride! This includes apples, bananas, avocados, cherries, peaches, strawberries, watermelons, and many others.
How is a standardization check done for fluorine?
Thus a standardization check is done every time fluorine is determined. With air-sensitive and/ or liquid samples digestion is done after sealing the sample in a Sn Capsule. Fluorine is a complex and labor intensive determination. It is important to note that the sample does not come into contact with glass through out the process.
How is fluorine tested in a micro bomb?
For fluorine testing, the appropriate size solid sample is weighed and inserted into a micro bomb. The bomb is then charged with the appropriate reagents to completely digest the fluorine compound. The apparatus is clamped shut and heated in a flame for 60 seconds. The contents are washed out and diluted.
How is fluorine determined in an elemental test?
Fluorine is a complex and labor intensive determination. It is important to note that the sample does not come into contact with glass through out the process. Aluminum and -OH are the only know interference. Strong base is circumvented in the dilution process.
How does fluorine react with all other elements?
It will react with nearly every other element. It is also the most electronegative of the elements, meaning that it attracts electrons towards itself. In standard conditions fluorine forms a gas made up of two fluorine atoms called a diatomic gas.
Thus a standardization check is done every time fluorine is determined. With air-sensitive and/ or liquid samples digestion is done after sealing the sample in a Sn Capsule. Fluorine is a complex and labor intensive determination. It is important to note that the sample does not come into contact with glass through out the process.
For fluorine testing, the appropriate size solid sample is weighed and inserted into a micro bomb. The bomb is then charged with the appropriate reagents to completely digest the fluorine compound. The apparatus is clamped shut and heated in a flame for 60 seconds. The contents are washed out and diluted.
Are there any materials that can contain fluorine?
Key word is “element”. There are many materials that can contain Fluorine, such as fluorinated metals, fluorinated graphene, and fluorine-based polymers. The key is that these materials have already reacted with fluorine and contain fluorine and can safely come in contact with the element without reacting. see more.
Fluorine is a complex and labor intensive determination. It is important to note that the sample does not come into contact with glass through out the process. Aluminum and -OH are the only know interference. Strong base is circumvented in the dilution process.