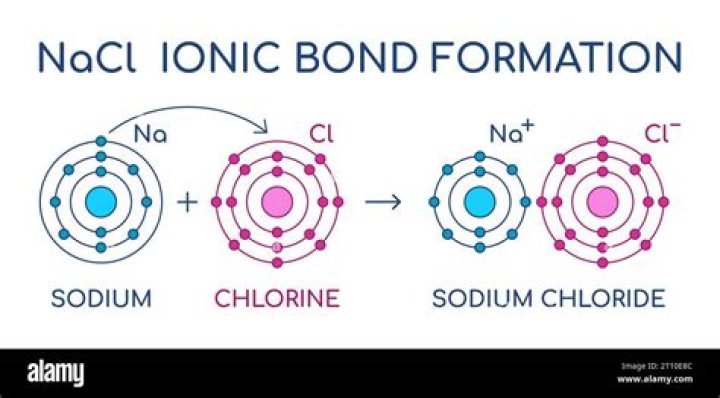
How is NaCl formation equation?
Answer: An example is the reaction in which sodium (Na) combines with chlorine (Cl 2 ) to form sodium chloride, or table salt (NaCl). … 2 Na ( s ) + Cl 2 ( g ) → 2 NaCl ( s ) 2.
Is NaCl an equation?
NaCl
Sodium chloride/Formula
How do you write an equation for the enthalpy of formation?
This equation essentially states that the standard enthalpy change of formation is equal to the sum of the standard enthalpies of formation of the products minus the sum of the standard enthalpies of formation of the reactants. and the standard enthalpy of formation values: ΔH fo[A] = 433 KJ/mol. ΔH fo[B] = -256 KJ/mol.
What is the full form of NaCl?
Chemical abbreviation for sodium chloride (table salt).
How do you get NaCl?
Fill a flask with 0.8 liters of water, weigh out the amount of sodium chloride you need, add it to the water and shake until it dissolves. To make a 1M solution, add 58.44 grams of salt; to make a 0.1M solution, add 5.84 grams; to make a 2M solution, add 116.88 grams and so on.
What is the formation reaction of CH3OH?
CH3OH and H2CO were formed as major products from the 100 eV electron-irradiated mixed CH4/H2O solid at 10 K. There found to be two pathways for the formation of methanol with about equal importance, i.e., the recombination reaction: CH3 + OH → CH3OH, and the insertion reaction: CH2+H2O → CH3OH.
What is a formation equation in chemistry?
1. A formation reaction is a reaction that produces one mole of a substance from its elements. Example: C(s) + O 2(g) → CO 2(g) 3. a) formation reaction.
Why is NaCl not nacl2?
The increase in the lattice energy that would result from forming an Na2+ ion can’t begin to compensate for the energy needed to break into the filled-shell configuration of the Na+ ion to remove a second electron. The reaction between sodium and chlorine therefore stops at NaCl.
What type of reaction is NaCl?
A synthesis reaction occurs when two or more reactants combine to form a single product. This type of reaction is represented by the general equation: A + B → AB. An example of a synthesis reaction is the combination of sodium (Na) and chlorine (Cl) to produce sodium chloride (NaCl).