How is aspartate Transcarbamoylase regulation?
ATCase is allosterically regulated by NTPs binding to the interface between adjacent regulatory subunits. Each allosteric site contains two nucleotide-specific subsites, A and B, that each bind one NTP and are bridged by a Mg2+ cation (or another divalent cation like Zn2+ or Ca2+).
What is the function of aspartate Transcarbamoylase?
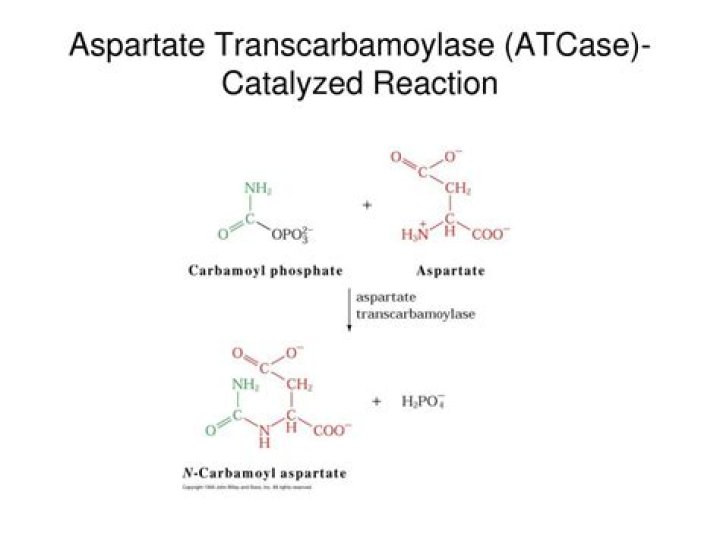
Aspartate transcarbamoylase (ATCase) catalyzes a key step of pyrimidine biosynthesis, the condensation of carbamoyl phosphate with aspartate to form N-carbamoylaspartate. The Escherichia coli enzyme has been extensively studied.
What type of modulator is aspartate Transcarbamoylase?
allosteric modulation
The enzyme is an archetypal example of allosteric modulation of fine control of metabolic enzyme reactions. ATCase does not follow Michaelis–Menten kinetics. Instead, it lies between its low-activity, low-affinity “tense” and its high-activity, high-affinity “relaxed” states.
What enzyme class is aspartate Transcarbamoylase?
allosteric inhibitor
Aspartate Transcarbamoylase Cytidine triphosphate (CTP) is an allosteric inhibitor representing a classic case of feedback inhibition whereby the end product of a biosynthetic pathway inhibits an enzyme catalyzing a reaction at the beginning of the pathway.
How does aspartate affect ATCase?
Aspartate transcarbamoylase (ATCase): Structure, role as an enzyme in a biosynthetic pathway and in regulation by feedback inhibition. Allosteric properties of ATCase. Once CbmP is bound, the affinity of the enzyme for aspartate is increased markedly, and aspartate shows a positive cooperative effect on catalysis.
How many binding sites does aspartate transcarbamylase have?
six active sites
Aspartate carbamoyltransferase (ATCase) is a large dodecameric enzyme with six active sites that exhibits allostery: its catalytic rate is modulated by the binding of various substrates at distal points from the active sites.
Is aspartate Transcarbamoylase involved in purine biosynthesis?
Aspartate Transcarbamoylase Adenosine triphosphate (ATP) is an allosteric activator, and together CTP and ATP act on ATCase to coordinate the rates of purine and pyrimidine nucleotide biosynthesis. The enzyme has the subunit composition c6r6, where c and r are catalytic and regulatory subunits, respectively.
What is CTP biology?
Cytidine triphosphate (CTP) is an allosteric inhibitor representing a classic case of feedback inhibition whereby the end product of a biosynthetic pathway inhibits an enzyme catalyzing a reaction at the beginning of the pathway. From: Encyclopedia of Biological Chemistry (Second Edition), 2013.
How do ATP and CTP affect the function of ATCase?
Cytidine triphosphate (CTP), which is an end product of the pyrimidine biosynthetic pathway, has a negative allosteric effect on ATCase activity, while adenosine triphosphate, ATP, has a positive allosteric effect. PALA has a Kd of about 10 nM for ATCase, meaning it binds the enzyme quite strongly.
What is the structure of ATCase?
ATCase consists of two catalytic trimers and three regulatory dimers that are completely separable units. It has been documented that the subunits can even combine when mixed together, reconstituting the enzyme.
What do you understand by Allosterism and cooperativity?
Allosterism describes the change in the affinity for binding of a ligand or substrate that is caused by the binding of another ligand away from the active site (allosteric = other site). • Allosterism is not the same as cooperativity. • Cooperativity creates the sigmoid curve.
What step controls the activity of both purine and pyrimidine synthesis?
Control of purine nucleotide synthesis has two phases. Control of the synthesis as a whole occurs at the amidotransferase step by nucleotide inhibition and/or [PRPP]. The second phase of control is involved with maintaining an appropriate balance (not equality) between ATP and GTP.