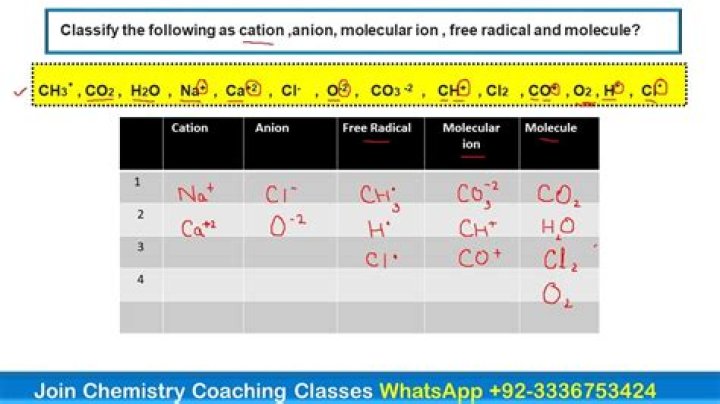
How do you write a cation formula?
The positive ion, called a cation, is listed first in an ionic compound formula, followed by the negative ion, called an anion. A balanced formula has a neutral electrical charge or net charge of zero….Examples of Ionic Compounds.
| Compound Name | sodium chloride |
|---|---|
| Formula | NaCl |
| Cation | Na+ |
| Anion | Cl- |
How do you write the formula for cations and anions?
When writing the formula for the ionic compound, the cation comes first, followed by the anion, both with numeric subscripts to indicate the number of atoms of each. Polyatomic ions are a set of covalently bonded atoms that have an overall charge, making them an ion.
What is the formula for anions?
Table of Common Anions
| Simple Anions | Formula |
|---|---|
| Oxide | O2- |
| Fluoride | F- |
| Sulfide | S2- |
| Chloride | Cl- |
What is formula unit in chemistry?
A formula unit in chemistry is the empirical formula of any ionic or covalent network solid compound used as an independent entity for stoichiometric calculations. Ionic compounds do not exist as individual molecules; a formula unit thus indicates the lowest reduced ratio of ions in the compound.
What is cation and anion?
Cations are positively-charged ions (atoms or groups of atoms that have more protons than electrons due to having lost one or more electrons). Anions are negatively-charged ions (meaning they have more electrons than protons due to having gained one or more electrons).
What is the formula of the compound containing Al3+ and OH ions?
Al(OH)3 is an ionic compound. Ionic compounds are named for the ions that are present, and prefixes aren’t used. The compound Al(OH)3 contains the aluminum ion (Al3+) and the hydroxide ion (OH−) . The name of this ionic compound is aluminum hydroxide.
What is the formula for Al3+ and CO32?
Aluminum carbonate | Al2(CO3)3 – PubChem.
What are anions and cations how are these anion and cation formed?
Cations (positively-charged ions) and anions (negatively-charged ions) are formed when a metal loses electrons, and a nonmetal gains those electrons. And all of them form an anion with a single negative charge. The VIA elements gain two electrons to form anions with a 2- charge.
Is formula units the same as moles?
1 mol of an atom, molecule or formula unit contains 6.02 x 1023 atoms, molecules or formula units.