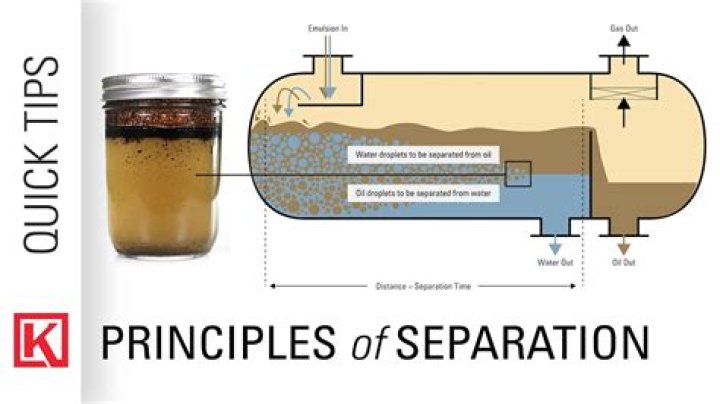
How do you separate emulsions?
Use gravity to separate your oil and water emulsion. Centrifuge the two substances together. The heavier liquid will reach the bottom first and stay there, while the lighter of the two will remain in a layer on the top. High G-forces are required.
Which method is most suitable for separating an emulsion?
Gravity separation is the most widely used method for oil emulsion separation. The elements in the well stream such as oil and water have different gravities. The density differences allow water to separate by gravity. With enough time in a non-turbulent state, the differing specific gravities will naturally separate.
What is it called when an emulsion separates?
An emulsion can be defined as a mixture of oily and watery liquids. To make an emulsion you need an emulsifier and force such as whisking and beating to break the oil droplets apart so they mix with the watery liquid. There are two types of emulsions.
How can you separate this mixture?
Summary
- Mixtures can be separated using a variety of techniques.
- Chromatography involves solvent separation on a solid medium.
- Distillation takes advantage of differences in boiling points.
- Evaporation removes a liquid from a solution to leave a solid material.
- Filtration separates solids of different sizes.
Why do emulsions separate?
How Does Emulsification Work? Two unlike substances won’t form an emulsion on their own—you need help, in the form of an emulsifier. The emulsifier coats the droplets, keeping them separate from each other, because when left to their own devices, the droplets will clump together, causing the emulsion to separate.
Can you fix broken emulsion?
Fixing any broken egg-based emulsion requires the same method: Create a new emulsion, then whisk the broken one into it. You can do this by placing a teaspoon of lemon juice (or water) in a clean bowl and adding a small amount of the broken emulsion, whisking to form another, stable emulsion.
What does a broken emulsion look like?
When you are adding the fat (for example the butter or olive oil), if you notice that the mixture begins to look grainy or curdled, then this is a good indication that the emulsion is about to break or separate.
What are the 5 ways to separate mixtures?
Some of the common methods of separating substances or mixtures are:
- Handpicking.
- Threshing.
- Winnowing.
- Sieving.
- Evaporation.
- Distillation.
- Filtration or Sedimentation.
- Separating Funnel.
How to troubleshoot problems with emulsions, emulsion separation?
How to troubleshoot problems with emulsions, emulsion separation and finding the right emulsifier for making creams and lotions HOMEABOUTFAQs INGREDIENTS All Ingredients Ingredients A-Z Active Ingredients Alpha & Beta Hydroxy Acids Anti-Aging Agents Anti-Dark Circles Agents Antioxidants Anti-Wrinkle Agents Hair Repair Agents Humectants Liposomes
How are oils and water mixed in an emulsion?
Emulsifier: An emulsifier is a special substance that is able to bind water molecules and oil molecules at the same time thereby mixing oils and water to a homogenous solution. Mechanical Force : By vigorous stirring the oil and water phase are broken up into fragments and small droplets forming a mixture held together by the emulsifier.
How is the emulsion of a droplet stabilized?
These molecules usually carry an electric charge and seek out the oil/water interface of the emulsified droplet. Here, the accumu- lated charges cause the emulsion to be stabilized through repulsion of the com- monly charged droplets.
How is the amount of emulsifier needed for a lotion?
The amount of emulsifier needed for a given emulsion is highly dependent on the droplet size (ie, surface area), the emulsifier efficiency, and the composition of the two phases.
How are solutions, mixtures and emulsions separated?
Learn the basics about separating solutions, mixtures and emulsions when learning about separation techniques as a part of chemical tests. A solution is a mixture of two or more substances in the same phase. It is formed when a solute dissolves in a solvent.
What kind of emulsion can be extracted from water?
These oils are defined as substances that can be extracted from water by hexane, carbon tetrachloride, chloroform, or fluorocarbons. In addition to oils, typical contaminants of these emulsions may be solids, silt, metal particles, emulsifiers, cleaners, soaps, sol- vents, and other residues.
These molecules usually carry an electric charge and seek out the oil/water interface of the emulsified droplet. Here, the accumu- lated charges cause the emulsion to be stabilized through repulsion of the com- monly charged droplets.
How is phase separation a sign of emulsion instability?
Phase separation is the most obvious sign of emulsion instability and involves the collision and coalescence of the oil phase droplets ultimately resulting in the visible appearance of separate oil and water phases. It is not realistic for a formulator to wait 1-2 years to check whether their emulsion is sufficiently stable.