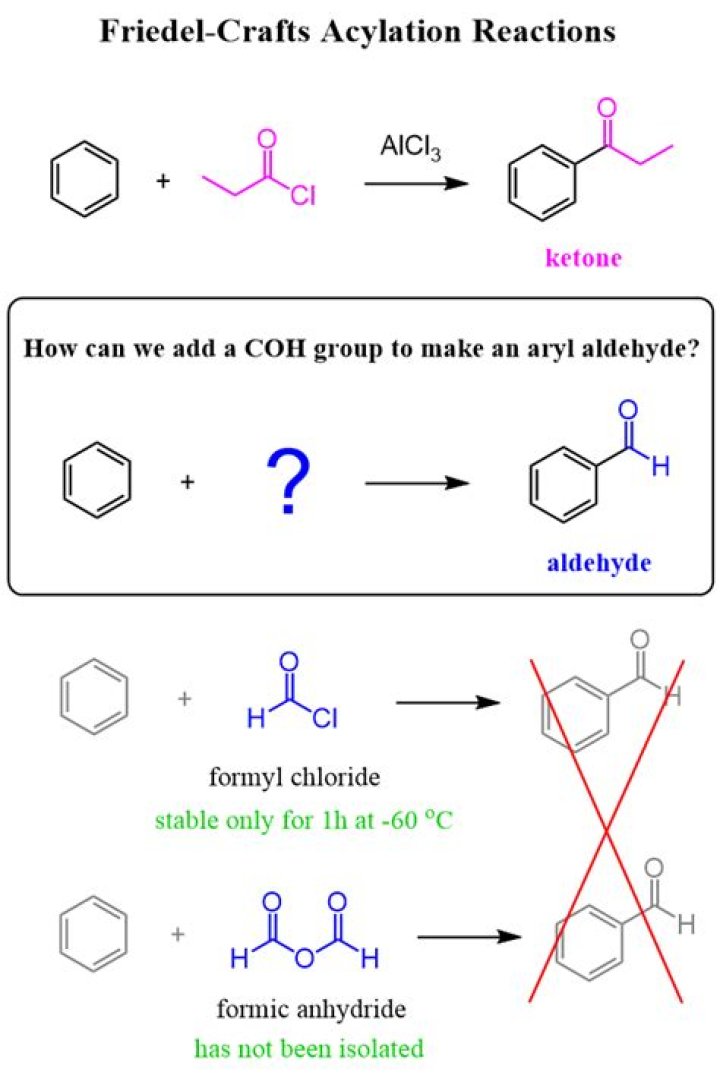
How do you make an aldehyde from an acid?
Acyl chloride/acid chloride undergoes hydrogenation in the presence of a catalyst such as barium sulfate (BaSO4) or Palladium (Pd) to form aldehydes. Aldehyde formation with this process is possible after the partial poisoning of the reaction by the addition of compounds such as sulfur or quinolone.
Can you reduce a carboxylic acid?
Carboxylic acids, acid halides, esters, and amides are easily reduced by strong reducing agents, such as lithium aluminum hydride (LiAlH 4).
Can nabh4 reduce carboxylic acid to aldehyde?
Although not as powerful as lithium aluminum hydride (LiAlH4), it is very effective for the reduction of aldehydes and ketones to alcohols. By itself, it will generally not reduce esters, carboxylic acids, or amides (although it will reduce acyl chlorides to alcohols).
Can LiAlH4 reduce carboxylic acid to aldehyde?
LiAlH4 is a strong, unselective reducing agent for polar double bonds, most easily thought of as a source of H-. It will reduce aldehydes, ketones, esters, carboxylic acid chlorides, carboxylic acids and even carboxylate salts to alcohols.
Which reagents can convert an aldehyde to a carboxylic acid?
Primary alcohols and aldehydes are normally oxidized to carboxylic acids using potassium dichromate(VI) solution in the presence of dilute sulfuric acid. During the reaction, the potassium dichromate(VI) solution turns from orange to green.
How are acid chlorides converted to aldehydes?
Acid chlorides can be reduced to aldehydes either by catalytic hydrogenation or by reaction with a metal hydride. Acid chlorides can also be converted to aldehydes with lithium tri(tert-butoxy)aluminum hydride. The reagent is prepared by reacting lithium aluminum hydride with three equivalents of tert-butyl alcohol.
How do carboxylic acids turn into alcohol?
Carboxylic acids can be converted to 1o alcohols using Lithium aluminum hydride (LiAlH4). Note that NaBH4 is not strong enough to convert carboxylic acids or esters to alcohols.
Does NaBH4 reduce epoxide?
Sodium borohydride is a relatively selective reducing agent. Ethanolic solutions of sodium borohydride reduce aldehydes and ketones in the presence of epoxides, esters, lactones, acids, nitriles, or nitro groups.
Does NaBH4 reduce carboxylic acid?
Standard organic chemistry texts discuss the lower re- activity of NaBH4 compared with lithium aluminum hy- dride, LiAlH4: whereas LiAlH4 reduces carboxylic acids to primary alcohols, NaBH4 does not reduce carboxylic acids.
How do you convert alcohol to aldehydes?
The primary alcohol is converted to aldehyde by the oxidation reaction using mild oxidizing reagent….Few mild oxidizing reagents used for the conversion of primary alcohol to aldehyde are as follows:
- Collins reagent: CrO3.2C5H5N.
- PCC: pyridinium chlorochromate.
- PDC: pyridinium dichromate (C5H5NH)22+Cr2O72−