How do you make a supersaturated salt solution?
Pour 8 oz. of water into a pan, and slowly add salt. When excess salt starts to rest at the bottom of the pan, move the pan to a burner to warm it. Stir the solution until the rest of the salt dissolves into the liquid.
How can you cause a supersaturated solution to crystallize?
The recrystallization of the excess dissolved solute in a supersaturated solution can be initiated by the addition of a tiny crystal of solute, called a seed crystal. The seed crystal provides a nucleation site on which the excess dissolved crystals can begin to grow.
What is an example of a supersaturated solution?
Examples of Supersaturated Solution Supersaturated solution contains more dissolved substances than a saturated solution. For example, 40g NaCl in 100ml H2O. The additional 4.0g NaCl remains undissolved.
What will happen if we cool a supersaturated solution?
The solid crystals will dissolve into the water in the hydrated crystals forming a supersaturated solution. If the sodium thiosulfate solution is cooled slowly the supersaturated solution will remain liquid. Placing a small crystal in the supersaturated solution will cause the liquid to turn solid.
What are the 2 ways to make a supersaturated solution?
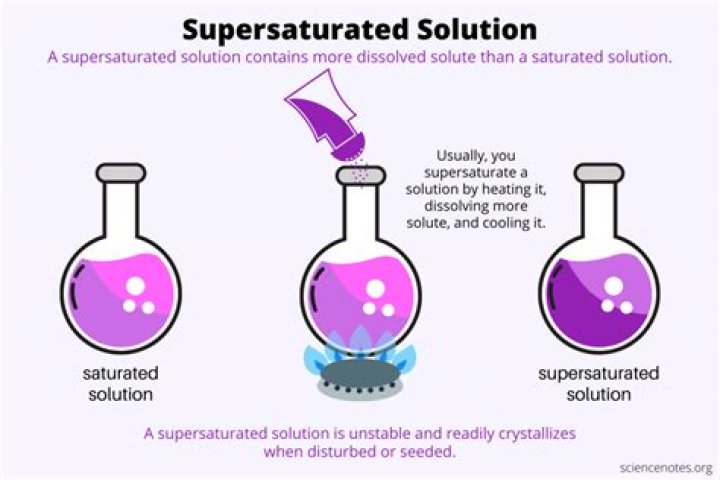
Supersaturated solutions are generally prepared by dissolving your compound in heated water. If you add sugar, for example, to water at 25 degrees Celsius, about 210 grams of sugar will dissolve per 100 mL of water.
How do you know if a solution is supersaturated?
It’s easy to tell if a solution is unsaturated, saturated, or supersaturated by adding a very small amount of solute. If the solution is unsaturated, the solute will dissolve. If the solution is saturated, it won’t. If the solution is supersaturated, crystals will very quickly form around the solute you’ve added.
What does a supersaturated solution look like?
Remember, a supersaturated solution will look just like an unsaturated solution with no solute settled at the bottom. However, there is more solute than the solvent can actually hold. Any slight change to the solution will cause all of the solute to come out.
What is an example of supersaturated solution?
A supersaturated solution is a more solute solution than can be dissolved by the solvent. The most popular example is sodium acetate which is supersaturated.
Is simple syrup a supersaturated solution?
What you create is a supersaturated sugar solution (aka simple syrup). It’s called “supersaturated” because it simply cannot hold any more sugar molecules. Now if you cover the bottom of a dish with this solution and add a sugar cube, the cube will get wet — but won’t dissolve.
What happens when supersaturated solution is cooled?
What happens when a supersaturated solution is cooled? The solid crystals in the hydrated crystals will dissolve into the bath, forming a supersaturated solution. When the solution for sodium thiosulfate is gradually cooled the super-saturated solution should remain liquid.
What happens when you agitate a supersaturated solution?
Supersaturated solutions are extremely unstable and will precipitate, or crystallize, upon addition of just one crystal of the sol- ute. Even slight shaking or agitation may be enough to cause crystallization to begin.
What is supersaturated solution give example?
A supersaturated solution is a more solute solution than can be dissolved by the solvent. If you haven’t learned what a solute / solvent is, the material that is dissolved in the solution, such as salts but not restricted to salts, is a solution. The most popular example is sodium acetate which is supersaturated.
How do you make a saturated Epsom salt solution?
Fill the container with water. Add some Epsom salts and stir. Watch as you’re stirring to see if all the salts are dissolving. If all the salts dissolve and disappear, add more salts and repeat. Once you have a layer of salt sitting on the bottom of your container, you know you have a saturated solution.
How long does it take to make Epsom salt crystals?
Epsom salt is primarily used for health reasons. People use it to make soft water for relaxing, treat constipation, and sore muscles. You, though, can use it to make Epsom salt crystals! Usually it takes several days to grow crystals. However, this super-easy recipe gives you a cup full of needle-like Epsom salt crystals in just a few hours!
How to prepare supersaturated salt water solutions?
Pour 8 oz. of water into a pan, and slowly add salt. When excess salt starts to rest at the bottom of the pan, move the pan to a burner to warm it. Stir the solution until the rest of the salt dissolves into the liquid. Slowly add more salt until a few crystals remain on the bottom of the pan. Remove the pan from the burner.
What’s the best way to store Epsom salt?
Grab a container with a lid that you’ll store your saturated Epsom salt solution in. A clear container makes it easy to see if you have undissolved salt crystals on the bottom. Fill the container with water. Add some Epsom salts and stir. Watch as you’re stirring to see if all the salts are dissolving.
How do you make supersaturated Epsom salt crystals?
To get a supersaturated solution you can either cool down the solution or let some of the water evaporate. To begin, make some Epsom salt crystals. These are easy to grow and you will begin to see crystals in a couple of hours. Start with one cup of warm distilled water (not boiling).
Do you have to use hot water to make Epsom salt?
You don’t have to use hot water when making your saturated Epsom salt solution but doing so will speed up the dissolving process. I do recommend using hot water if you’re making the solution right before you’re going to use it. But then it’ll cool over time and be room temperature every other time you use it.
How do you make a supersaturated solution of water?
However, if you heat the water up to 80 degrees Celsius, the same amount of water will now dissolve 360 grams of sugar. To make a supersaturated solution, make a saturated solution of sugar by adding 360 grams of sugar to 100 mL of water at 80 degrees Celsius.
What makes an epsom salt solution a saturated solution?
Additional solute will not dissolve in a saturated solution. In our case, the solute is Epsom salts and the solvent is water. So a saturated Epsom salt solution will have the highest possible number of magnesium ions per volume of water.