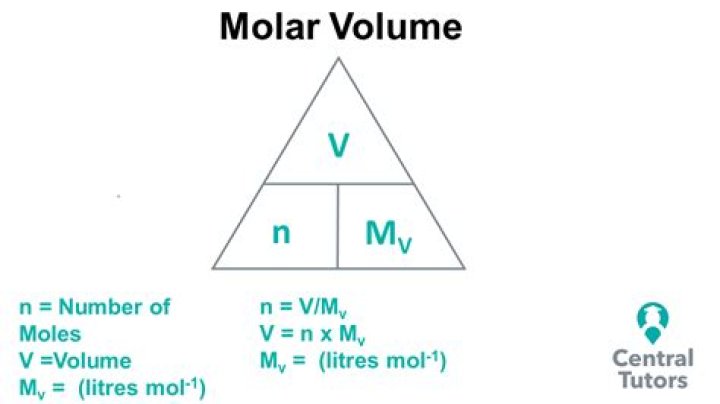
How do you find the molar volume?
At standard Temperature and Pressure (STP) the molar volume (Vm) is the volume occupied by one mole of a chemical element or a chemical compound. It can be calculated by dividing the molar mass (M) by mass density (ρ). Molar gas volume is one mole of any gas at a specific temperature and pressure has a fixed volume.
How much is the molar volume?
Example. The molar volume of an ideal gas at STP is 22.4 L/mol. Helmenstine, Anne Marie, Ph. D.
What is the molar volume of n2?
So, at STP, or Standard Temperature and Pressure, one mole of any ideal gas occupies exactly 22.7 L – this is known as the molar volume of a gas at STP. This means that if 28 g of nitrogen gas occupy 22.7 L at STP, then you can say that the molar mass of nitogen gas is 28 g/mol .
What is molar volume Class 9?
The Molar Volume, represented by V m, is the volume occupied by one mole of a substance which can be a chemical element or a chemical compound at Standard Temperature and Pressure (STP). It can be calculated by dividing Molar mass (M) by mass density (ρ).
What is molar volume 11th?
The molar volume is that the volume occupied by one mole of a substance which may be an element or a compound at degree Centigrade and Pressure (STP).
What is molar volume given example?
In simple words, Molar volume is the Volume occupied by one mole of any substance at a given temperature and pressure. Example: A sample of pure helium gas occupies a volume of 6.8 L at 0°C and 100 kPa.
What units are molar volume in?
It has the SI unit cubic meters per mole (m3/mol), and often practical to use the units cubic decimeters per mole (dm3/mol) for gases and cubic centimeters per mole (cm3/mol) for liquids and solids.
What is the molar volume of h2?
When the reaction between the metal and the acid is conducted in a eudiometer, the volume of the hydrogen gas produced can be easily determined. According to Avogadro’s law, the volume of one mole of any gas at Standard Temperature and Pressure (STP = 273 K and 1 atm) is 22.4 L.
What is N2 STP?
Density at STP (g/L) Molar Mass/Density (L/mole) N2, nitrogen gas. 28.014. 1.25.
What is molar volume in Class 11?
What is gram molar volume Class 11?
The volume occupied by 1 mole of a substance is called gram molar volume. the gram molar volume of perfect gas is 22.4L at STP.
What is molar volume of a gas Class 10?
Molar volume of a gas is defined as the volume of one mole of the gas. Thus, the molar volume is also the volume occupied by 6.02 x 1023 particles of gas. The molar volume of any gas is 22.4 dm3 mol-1 at STP or 24 dm3 mol-1 at room conditions.