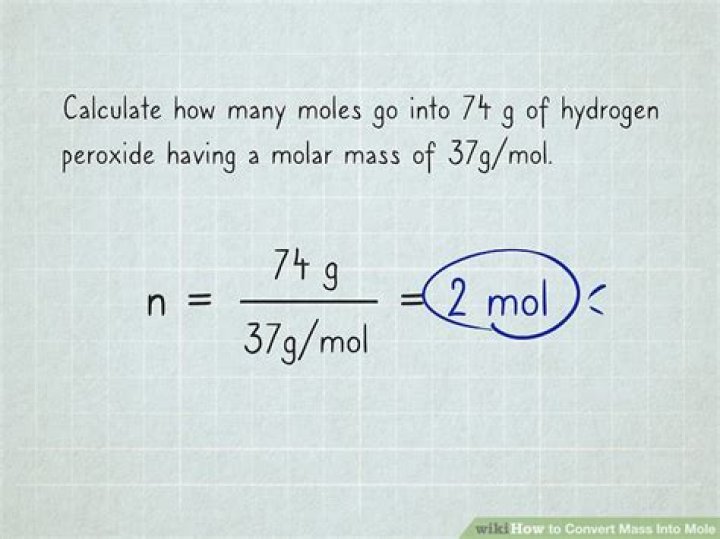How do you convert mass to molecules from moles?
A substance’s molar mass is calculated by multiplying its relative atomic mass by the molar mass constant (1 g/mol). The molar mass constant can be used to convert mass to moles. By multiplying a given mass by the molar mass, the amount of moles of the substance can be calculated.
How do you convert moles to molecules in chemistry?
Take the number of particles and divide them by Avogadro’s number. To convert moles to particles (atoms or molecules): Take the number of moles and multiply them by Avogadro’s number.
How did you convert moles to atoms or molecules )?
Avogadro’s number is a very important relationship to remember: 1 mole = 6.022×1023 6.022 × 10 23 atoms, molecules, protons, etc. To convert from moles to atoms, multiply the molar amount by Avogadro’s number. To convert from atoms to moles, divide the atom amount by Avogadro’s number (or multiply by its reciprocal).
How do you convert moles to particles?
From moles to particles (atoms, molecules, or formula units): Multiply your mole value by Avogadro’s number, 6.02 × 1023.
How do you find the molar mass of a molecule?
Determine the mass of the substance, and its molar mass. Divide the given mass by its molar mass to get moles, then multiply times 6.022×1023molecules1mol .
How many molecules are in a mole?
The mole is represented by Avogadro’s number, which is 6.022 × 1023 atoms or molecules per mol.
How do you find moles in chemistry?
If you want to know how many moles of a material you have, divide the mass of the material by its molar mass. The molar mass of a substance is the mass in grams of one mole of that substance. This mass is given by the atomic weight of the chemical unit that makes up that substance in atomic mass units (amu).
How do you convert moles to joules?
Since 1 mole = 6.02214076×1023 particles (atoms, molecules, ions etc.), 1 joule per mole is equal to 1 joule divided by 6.02214076×1023 particles, 1.66054×10−24 joule per particle.
How do you convert MOL to molecules?
Converting between Moles and Atoms
- Avogadro’s number is a very important relationship to remember: 1 mole = 6.022×1023 6.022 × 10 23 atoms, molecules, protons, etc.
- To convert from moles to atoms, multiply the molar amount by Avogadro’s number.