How can ketones be reduced to alkanes?
The reduction of aldehydes and ketones to alkanes. Condensation of the carbonyl compound with hydrazine forms the hydrazone, and treatment with base induces the reduction of the carbon coupled with oxidation of the hydrazine to gaseous nitrogen, to yield the corresponding alkane.
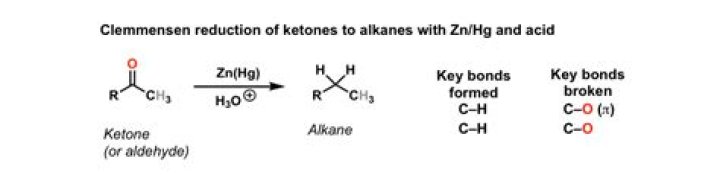
What reagent converts ketones to alkanes?
Clemmensen reduction is a chemical reaction described as a reduction of ketones (or aldehydes) to alkanes using zinc amalgam and concentrated hydrochloric acid.
How do they remove Thioacetal?
Among the known methods of removal of thioacetal protecting groups2, the one using cerium (IV) ammonium nitrate (CAN) has the advantages of a fast reaction with easy work-up and good to very good yields3. This method has been widely applied for preparative purposes using four equivalents of the cerium salt.
What does NI H2 reduce?
It is typically used in the reduction of compounds with multiple bonds, such as alkynes, alkenes, nitriles, dienes, aromatics and carbonyl-containing compounds. Additionally, Raney nickel will reduce heteroatom-heteroatom bonds, such as hydrazines, nitro groups, and nitrosamines.
What can reduce a ketone?
Aldehydes and Ketones are reduced by most reducing agents. Sodium borohydride and lithium aluminumhydride are very common reducing agents. Ketones and Aldehydes can also be reduced to the respective alkanes.
What reagent gets rid of ketone?
The relatively weak reducer sodium borohydride is typically used for reducing ketones and aldehydes because unlike lithium aluminum hydride, it tolerates many functional groups (nitro group, nitrile, ester) and can be used with water or ethanol as solvents.
Which of the following reagent convert ketone into alkene?
Wittig reactions are most commonly used to convert aldehydes and ketones to alkenes….
| Wittig reaction | |
|---|---|
| Reaction type | Coupling reaction |
| Reaction | |
| aldehyde or ketone + triphenyl phosphonium ylide ↓ alkene + triphenylphosphine oxide | |
| Conditions |
What conditions will turn a Thioacetal into the corresponding carbonyl?
Thioacetals have been traditionally converted into carbonyls by hydrolysis with heavy-metal reagents such as mercury(II) salts, or by oxidation, and these approaches were discussed in chapter 3.01.
What catalysts are used in the Perkin reaction?
Q2: What catalysts are used in the Perkin reaction? A2: The alkali salt of the acid anhydride is used as a catalyst. However, other bases can be used for this purpose.
Does H2 PD reduce Ester?
Reduce aldehyde or ketone to alcohol: use H2 with Raney nickel iv. Reduce acid chloride to aldehyde: use a partially deactivated Pd catalyst (this is the Rosenmund reduction) v. It will not reduce an acid or an ester.
Does H2 NI reduce COOH?
No ,hydrogen and Raney Nickel cannot reduce carboxyllic acids,esters , amides but they can reduce aldehydes ketones,alkenes ,alkynes ,cyanides,acidchlorides etc.