How can esters be synthesized from alcohols?
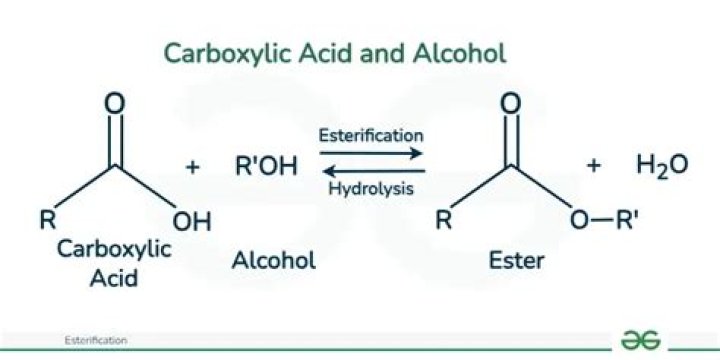
Alcohols can combine with many kinds of acids to form esters. The reaction, called Fischer esterification, is characterized by the combining of an alcohol and an acid (with acid catalysis) to yield an ester plus water. Under appropriate conditions, inorganic acids also react with alcohols to form esters.
How do you oxidize alcohol into ester?
Alcohols and aldehydes can be oxidized to the corresponding methyl esters by reaction with methanol in the presence of crotononitrile as a hydrogen acceptor using a catalyst combination of Ru(PPh3)3(CO)H2 with xantphos.
Which compound reacts with an alcohol to form an ester?
Description: When a carboxylic acid is treated with an alcohol and an acid catalyst, an ester is formed (along with water). This reaction is called the Fischer esterification. Notes: The reaction is actually an equilibrium. The alcohol is generally used as solvent so is present in large excess.
Can esters be formed from secondary alcohols?
An unexpected new reaction pathway, from which the secondary alcohols can be directly oxidized to corresponding esters, was found.
How are ethers formed?
There are two primary reactions to generate ethers: either by Dehydration of Alcohols or by the Williamson Synthesis. As stated previously, alkoxides are created by reacting an alcohol with metallic sodium or potassium, or a metal hydride, such as sodium hydride (NaH).
How do you make alcohol into ether?
At 110º to 130 ºC an SN2 reaction of the alcohol conjugate acid leads to an ether product. At higher temperatures (over 150 ºC) an E2 elimination takes place. In this reaction alcohol has to be used in excess and the temperature has to be maintained around 413 K.
Do esters react with alcohol?
When in ester is placed in a large excess of an alcohol along with presence of either an acid or a base there can be an exchange of alkoxy groups. Since both the reactants and the products are an ester and an alcohol, the reaction is reversible and the equilibrium constant is close to one.
How are esters formed from ether?
The -OH groups on a monosaccharide can be readily converted to esters and ethers. Esterfication can be done with an acid chloride (Section 21.4) or acid anhydride (Section 21.5), while treatment with an alkyl halide by a Williamson ether synthesis (Section 18.2) leads to the ether.
How is an ester obtained from alcohol and phenol?
Phenols react with carboxylic acids so slowly that the reaction is unusable for preparation purposes. Esters are produced when carboxylic acids are heated with alcohols in the presence of an acid catalyst. The catalyst is usually concentrated sulphuric acid. The esterification reaction is both slow and reversible.
How are ethers formed from alcohols?
Bimolecular dehydration In the presence of acid, two molecules of an alcohol may lose water to form an ether. In practice, however, this bimolecular dehydration to form an ether competes with unimolecular dehydration to give an alkene. Dehydration is used commercially to produce diethyl ether.