How are ketones reduced?
How are ketones reduced?
The reduction of aldehydes and ketones by sodium tetrahydridoborate. The reaction is carried out in solution in water to which some sodium hydroxide has been added to make it alkaline. The reaction produces an intermediate which is converted into the final product by addition of a dilute acid like sulphuric acid.
What type of reaction is reduction of a ketone?
Clemmensen reduction is a chemical reaction described as a reduction of ketones (or aldehydes) to alkanes using zinc amalgam and concentrated hydrochloric acid.
What reagent is used to reduce ketones to alcohols?
LiAlH4 and NaBH4 are both capable of reducing aldehydes and ketones to the corresponding alcohol.
Does ketone undergo reduction?
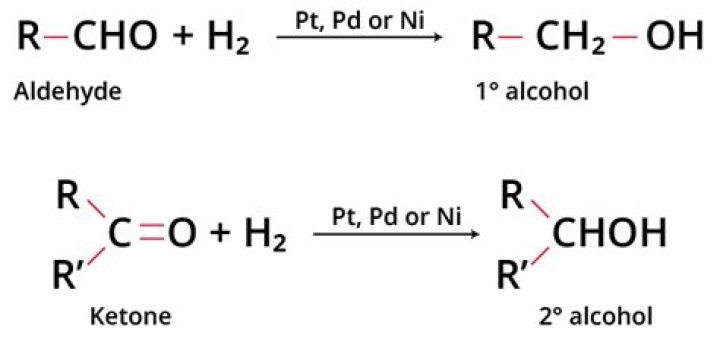
Aldehydes and ketones can undergo reduction process for the formation of either primary alcohol or secondary alcohol with the help of reagents, sodium borohydride (NaBH4) or lithium aluminium hydride (LiAlH4). Aldehydes and ketones can also form alcohol by the process of catalytic hydrogenation.
Which reagent does not reduce ketone?
Sodium borohydride or Lithium Aluminium hydride.
Can H2 reduce ketone?
H2 Ni are strong reducing agents which reduces aldehydes to primary alcohols,ketones to secondary alcohols and carboxylic acid and its derivatives to primary alcohols.
What happens when you oxidize a ketone?
Ketone oxidation implies the rupture of a C-C bond. If it is energic (KMnO4, K2Cr2O7) two carboxylic groups will be produced. If it is smooth (Baeyer-Villiger oxidation), an ester is produced that, once it is hydrolized, gives rise to a carboxylic acid and an alcohol.
How do you reduce ketones in alcohol?
Reduction of other aldehydes gives primary alcohols. Reduction of ketones gives secondary alcohols. The acidic work-up converts an intermediate metal alkoxide salt into the desired alcohol via a simple acid base reaction.
Which reagent can be used to reduce a ketone quizlet?
LAH is a strong reducing agent and will reduce Ketones, Aldehydes and Esters as well as Carboxylic acids.
Which reducing reagents are used for reduction of aldehydes and ketone?
The relatively weak reducer sodium borohydride is typically used for reducing ketones and aldehydes because unlike lithium aluminum hydride, it tolerates many functional groups (nitro group, nitrile, ester) and can be used with water or ethanol as solvents.
Do ketones reduce tollens reagent?
Ketones do not reduce Tollens’ reagent (except alpha diketones (i.e. two ketone functionalities on the adjacent atoms) and alpha hydroxy ketones (keto and alcohol functional groups on adjacent atoms) which can tautomerize into aldehydes via keto-enol tautomerism in an alkaline medium).
Which reagent is used to reduce acid or ester?
Lithium aluminum hydride LiAlH4 is a strong, unselective reducing agent for polar double bonds, most easily thought of as a source of H-. It will reduce aldehydes, ketones, esters, carboxylic acid chlorides, carboxylic acids and even carboxylate salts to alcohols.



