How are chemicals arranged in chemical families?
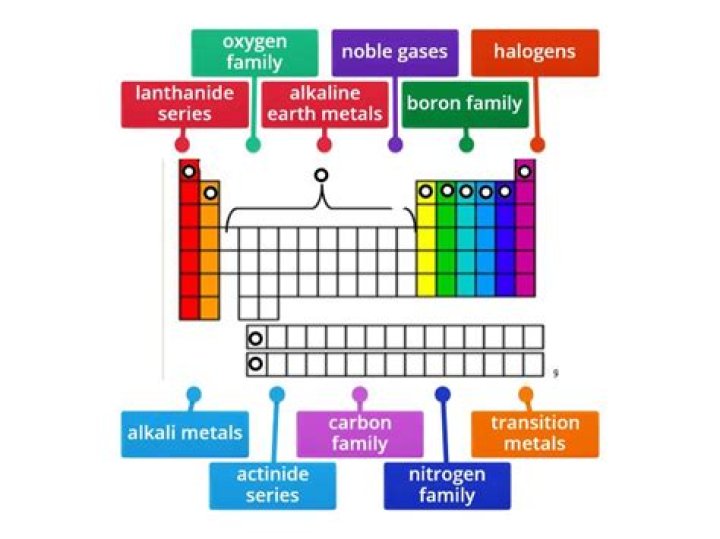
The vertical columns on the periodic table are called groups or families because of their similar chemical behavior. All the members of a family of elements have the same number of valence electrons and similar chemical properties. The horizontal rows on the periodic table are called periods.
What are the 4 chemical families?
Families in the periodic table
- The IA family is made up of the alkali metals. In reactions, these elements all tend to lose a single electron.
- The IIA family is made up of the alkaline earth metals.
- The VIIA family is made up of the halogens.
- The VIIIA family is made up of the noble gases.
What is a chemical family in chemistry?
In chemistry, a group (also known as a family) is a column of elements in the periodic table of the chemical elements. There are 18 numbered groups in the periodic table; the f-block columns (between groups 2 and 3) are not numbered. Groups may also be identified using their topmost element, or have a specific name.
How do you combine chemical structures?
A compound is a unique substance that forms when two or more elements combine chemically. Compounds form as a result of chemical reactions. The elements in compounds are held together by chemical bonds. A chemical bond is a force of attraction between atoms or ions that share or transfer valence electrons.
What family combines with metals to make salts?
alkali metals
Halogen Family The elements in this family are fluorine, chlorine, bromine, iodine, and astatine. They are the most active non-metals. They are never found free in nature. They react with alkali metals to form salts.
What is the symbol for magnesium?
Mg
Magnesium/Symbol
How are valence electrons related to chemical families?
Electrons in the outermost shell are called valence electrons, because it is their interactions that determine the chemical properties of an element. The columns that were set up to group elements by similar chemical properties turn out to be the exact same columns defined by the number of valence electrons.
How do you combine two molecules?
To merge two molecules, you can add a bond betwwen two atoms, one from each molecule. The drawback is that this method will not align properly the two parts of your new molecule. If one of the molecule has two or more atoms at the same position than an atom of the second molecule, the result is not easily preditable.
How are combinations of different atoms held together?
Every combination of atoms is a molecule. There are two main types of chemical bonds that hold atoms together: covalent and ionic/electrovalent bonds. Atoms that share electrons in a chemical bond have covalent bonds. An oxygen molecule (O2) is a good example of a molecule with a covalent bond.
Which group is known as noble gas family?
Group 18
noble gas, any of the seven chemical elements that make up Group 18 (VIIIa) of the periodic table. The elements are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn), and oganesson (Og).
Which 2 families of elements often combine to form salts?
Group 17: Halogens The two elements combine to form a salt. For example, the halogen chlorine (Cl) and the alkali metal sodium (Na) react to form table salt, or sodium chloride (NaCl).