Does the heavy chain have a variable region?
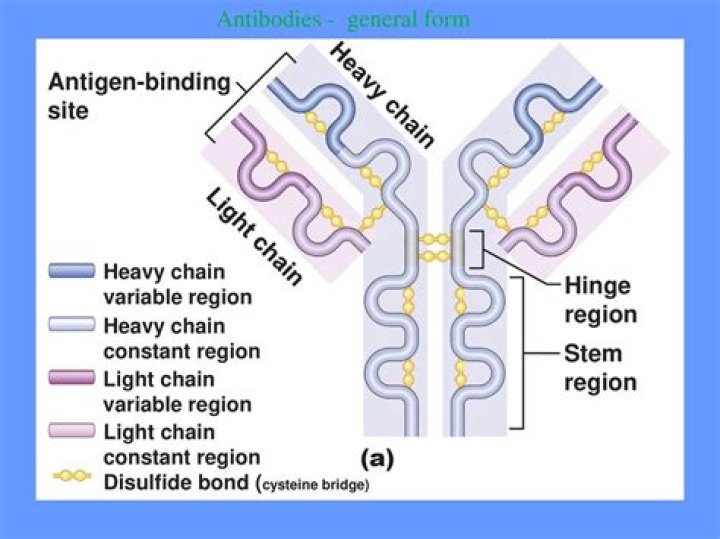
Each heavy chain has two regions, the constant region and the variable region. The constant region is identical in all antibodies of the same isotype, but differs in antibodies of different isotypes. The variable region of each heavy chain is approximately 110 amino acids long and is composed of a single Ig domain.
Which region is only present in heavy chain?
Each heavy chain has two regions: a constant region (which is the same for all immunoglobulins of the same class but differs between classes). Heavy chains γ, α and δ have a constant region composed of three tandem (in a line next to each other) immunoglobulin domains but also have a hinge region for added flexibility.
What contributes to heavy chain CDR3 length?
This region varies the most in length because it is constructed from several components. The heavy chain CDR3 is formed by amino acid residues encoded by a variable (VH) gene segment, diversity (D) gene segment, and joining (JH) gene segment.
What is the heavy chain of IgG?
IgG antibodies are large molecules, having a molecular weight of approximately 150 kDa, composed of two different kinds of polypeptide chain. One, of approximately 50 kDa, is termed the heavy or H chain, and the other, of 25 kDa, is termed the light or L chain (Fig. 3.2).
What is a variable region?
variable region –> V region. (Science: molecular biology) Those regions in the amino acid sequence of both the heavy and the light chains of immunoglobulins where there is considerable sequence variability from one immunoglobulin to other of the same class, in contrast to constant sequence (C) regions.
What is the variable vs constant region?
regions, called constant (C) and variable (V). These regions are distinguished on the basis of amino acid similarity—that is, constant regions have essentially the same amino acid sequence in all antibody molecules of the same class (IgG, IgM, IgA, IgD, or IgE), but the amino acid sequences of the variable…
How many heavy chains does IgA?
Like all Igs, IgA molecules are made up of pairings of two identical heavy chains (α-chains in the case of IgA) and two identical light chains.
How many CDR are in an antibody?
A single antibody molecule has two antigen receptors and therefore contains twelve CDRs total. There are three CDR loops per variable domain in antibodies. Sixty CDRs can be found on a pentameric IgM molecule.
Why is CDR3 important?
Because of its great potential for diversity, the immunoglobulin heavy-chain complementarity-determining region 3 (HCDR3) is taken as an antibody molecule’s most important component in conferring binding activity and specificity.
What is a heavy chain in an antibody?
A heavy-chain antibody is an antibody which consists only of two heavy chains and lacks the two light chains usually found in antibodies. This observation has led to the development of a new type of antibody fragments with potential use as drugs, so-called single-domain antibodies.
What is the function of the heavy chain?
Heavy chains may contain a transmembrane domain that allows for Ig to be expressed on the surface of B cells. They allow for antigen-specific binding and subsequent activation of B lymphocytes. Importantly, antigen binding by the antibody is not sufficient to induce a cellular activation signal.