Does Fructopyranose exist?
Does Fructopyranose exist?
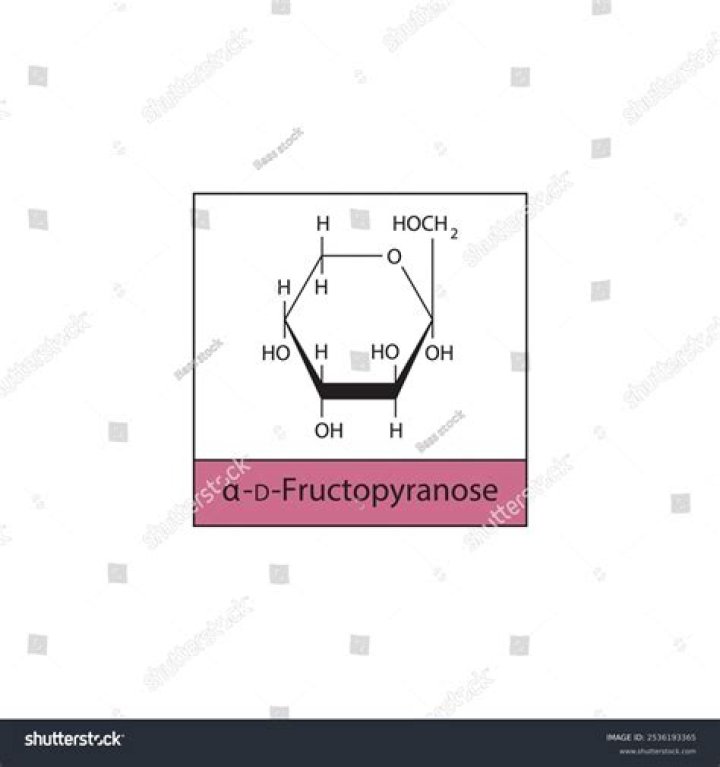
group at C2 and form a cyclic hemiketal. The product is called fructopyranose in analogy with the six-membered ring of pyran. The formation of a cyclic ketal generates a new stereocentre at C2. Thus, both fructofuranose and fructopyranose exist in two stereoisomeric forms.
What is meant by D fructose?
D-fructopyranose is a fructopyranose having D-configuration. It has a role as a sweetening agent. It is a fructopyranose, a D-fructose and a cyclic hemiketal. ChEBI. A monosaccharide in sweet fruits and honey that is soluble in water, alcohol, or ether.
What is the difference between Fructopyranose and Fructofuranose?
Fructopyranose is a structure which is analogous to the cyclic structure called as pyran, which is a 6 carbon membered ring. Fructofuranose is a structure which is analogous to the cyclic structure called furan which is a 5 membered ring.
How Fructopyranose is formed?
Fructose in aqueous solution forms a six-membered cyclic hemiketal called fructopyranose when the hydroxyl oxygen on carbon #6 attacks the ketone carbon (carbon #2, the anomeric carbon in fructose).
What does fructose look like?
Pure, dry fructose is a sweet, white, odorless, crystalline solid, and is the most water-soluble of all the sugars. Fructose is found in honey, tree and vine fruits, flowers, berries, and most root vegetables.
Is fructose good or bad?
Fructose is only harmful in large amounts, and it’s difficult to get excessive amounts of fructose from fruit. Evidence suggests that fructose can cause harm when consumed in excess. However, there is not enough fructose in fruit to cause concern.
What is difference between alpha and beta fructose?
If the OH group is pointing “down”, it is alpha. If the OH group points “up”, it is beta. The L-isomers of fructose are the corresponding mirror images of the D-isomers. If a group ( OH or CH2OH ) is “up” in one isomer, it is “down” in the other.