Does BCL-2 bind to Bax?
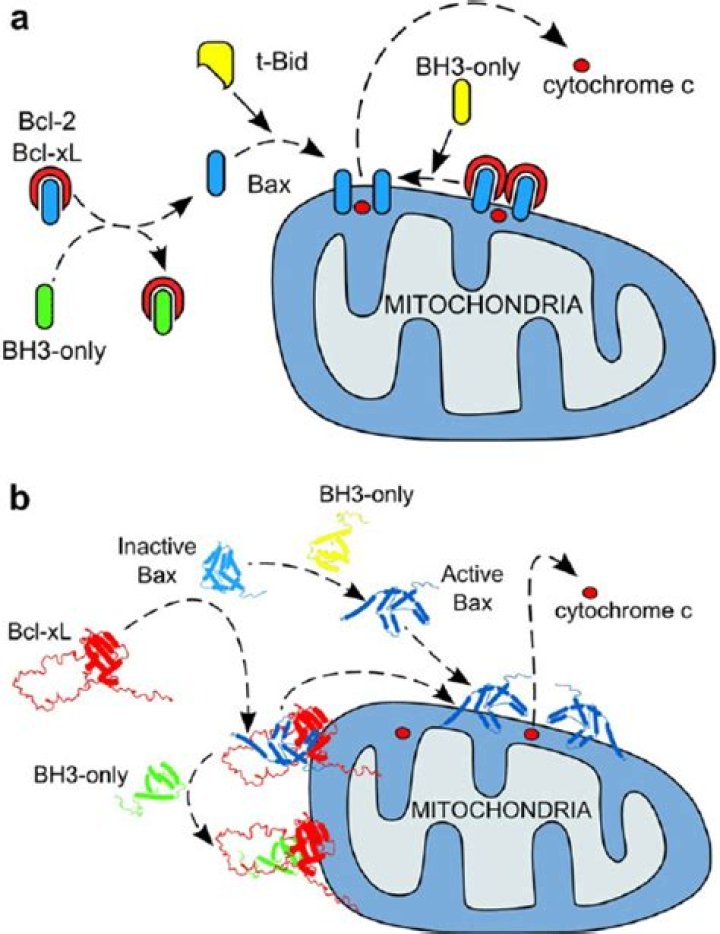
The distinctive feature of the direct activation model is that a BH3 protein is required to directly bind and to activate the Bcl-2 multihomology region proapoptotic proteins, Bax and Bak.
Is Bax a Bcl-2 family member?
The BCL-2 family is divided into three groups based on their primary function (1) anti-apoptotic proteins (BCL-2, BCL-XL, BCL-W, MCL-1, BFL-1/A1), (2) pro-apoptotic pore-formers (BAX, BAK, BOK) and (3) pro-apoptotic BH3-only proteins (BAD, BID, BIK, BIM, BMF, HRK, NOXA, PUMA, etc.).
What is the Bax pathway?
BAX is believed to interact with, and induce the opening of the mitochondrial voltage-dependent anion channel, VDAC. This results in the release of cytochrome c and other pro-apoptotic factors from the mitochondria, often referred to as mitochondrial outer membrane permeabilization, leading to activation of caspases.
Does BCL-2 inhibit Bax?
Bcl-2 inhibits Bax translocation from cytosol to mitochondria during drug-induced apoptosis of human tumor cells. Cell Death Differ. 2000 Jan;7(1):102-11. doi: 10.1038/sj.
What do Bak and Bax do?
Bax and Bak are two nuclear-encoded proteins present in higher eukaryotes that are able to pierce the mitochondrial outer membrane to mediate cell death by apoptosis. Thus, organelles recruited by nucleated cells to supply energy can be recruited by Bax and Bak to kill cells.
How is the Bax gene activated?
Facts. Bax and Bak are activated by BH3-only proteins and inhibited by prosurvival Bcl-2 proteins via direct interactions. Bax and Bak undergo major conformation changes during transition from inactive monomers to activated oligomers.
What is the function of BCL-2 proteins?
BCL-2 family proteins are the regulators of apoptosis, but also have other functions. This family of interacting partners includes inhibitors and inducers of cell death. Together they regulate and mediate the process by which mitochondria contribute to cell death known as the intrinsic apoptosis pathway.
What do Bax and Bak do?
How does Bax get activated?
Bax and Bak are activated by BH3-only proteins and inhibited by prosurvival Bcl-2 proteins via direct interactions. Bax and Bak undergo major conformation changes during transition from inactive monomers to activated oligomers.
How does Bcl-2 inhibit Bax?
Bcl-2 inhibits apoptosis by regulating the release of cytochrome c and other proteins from mitochondria. Thus, Bcl-2 functions as an inhibitor of mitochondrial permeabilization by changing conformation in the mitochondrial membrane to bind membrane-inserted Bax monomers and prevent productive oligomerization of Bax.