Does BaSO4 form a precipitate?
Since BaSO4 is a precipitate, it does not need to be split up into ions. These are called spectator ions, and do not take part in the reaction.
What is the Colour of BaSO4 precipitate?
The precipitate of barium sulphate is white in colour as the salt does not release any free electrons that undergo transitions in the visible energy range.
Why does BaSO4 form a precipitate?
BaSO4, HgSO4 and PbSO4 are insoluble. A solution of barium chloride is mixed with a solution of potassium sulfate and a precipitate forms. Because it is insoluble in water we know that it is the precipitate. As all of the other substances are soluble in water we can rewrite the equation.
What type of precipitate is BaSO4?
The reaction of barium sulfate in water is an ionization reaction: solvated ions (ions in solution) are formed from an insoluble precipitate.
How do you identify a precipitate?
An ionic solution is when the ions of a compound have dissociated in an aqueous solution. A reaction happens when you mix two aqueous solutions. This is when you find out if a precipitate will form or not. A precipitate forms if the product of the reaction of the ions is insoluble in water.
How do you know what the precipitate is?
If the rules state that an ion is soluble, then it remains in its aqueous ion form. If an ion is insoluble based on the solubility rules, then it forms a solid with an ion from the other reactant. If all the ions in a reaction are shown to be soluble, then no precipitation reaction occurs.
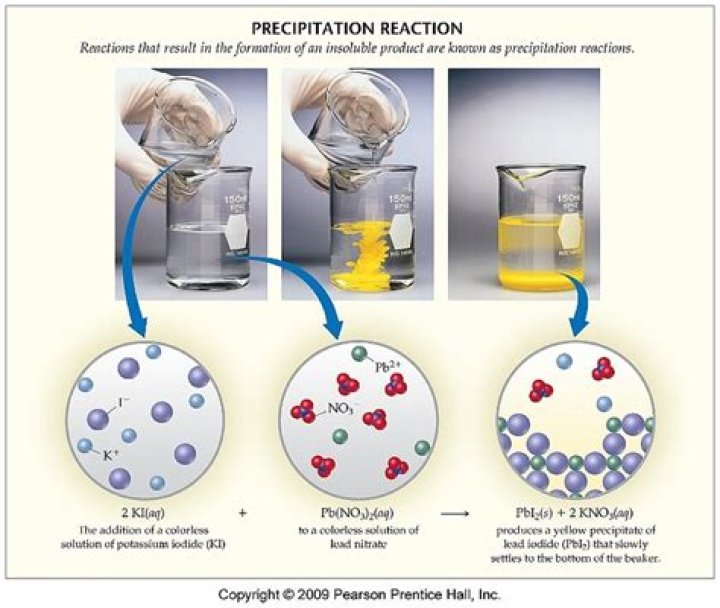
What is the yellow precipitate?
The formation of a precipitate is an indication of a chemical reaction. A yellow precipitate of solid lead (II) iodide forms immediately when solutions of lead (II) nitrate and potassium iodide are mixed.
Is precipitate of baso4 soluble in water and what is its Colour?
Barium sulfate (or sulphate) is the inorganic compound with the chemical formula BaSO4. It is a white crystalline solid that is odorless and insoluble in water.
Is BaSO4 aqueous or solid?
Barium sulfate (or sulphate) is the inorganic compound with the chemical formula BaSO4. It is a white crystalline solid that is odorless and insoluble in water. It occurs as the mineral barite, which is the main commercial source of barium and materials prepared from it.
What is the compound for BaSO4?
BaSO4
Barium sulfate/Formula
Which of the following defines a precipitate?
In chemistry, a precipitate is an insoluble solid that emerges from a liquid solution. The emergence of the insoluble solid from solution is called precipitation. Often the precipitate emerges as a suspension. Precipitates can also form when the temperature of a solution is lowered.
How do you identify a precipitation reaction?